Chapter 24: Q33P (page 1298)
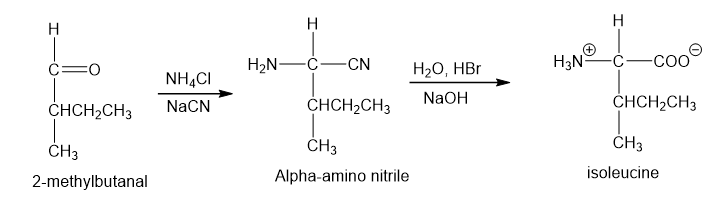
Show how you would use the Strecker synthesis to make isoleucine. What stereochemistry would you expect in your synthetic product?
Short Answer
Learning Materials
Features
Discover
Chapter 24: Q33P (page 1298)
Show how you would use the Strecker synthesis to make isoleucine. What stereochemistry would you expect in your synthetic product?

All the tools & learning materials you need for study success - in one app.
Get started for free
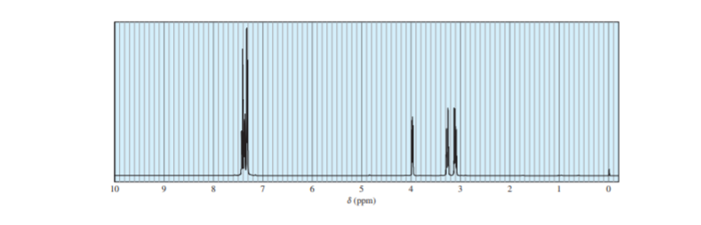
A student took the proton NMR spectrum of phenylalanine in D2Osolution, and had the instrument suppress the DOHsolvent peak. The spectrum is shown below. The integrated relative areas of the peaks are 5:1:1:1.
(a) Draw the structure of phenylalanine as it exists in D2Osolution. (There is a large excess of D2O, and any exchangeable protons in phenylalanine will exchange with the solvent.)
(b) Assign the peaks in the spectrum to the protons in the structure.
(c) Why don’t we see the -NH2or -COOHprotons in the spectrum?
(d) What is the relationship between the two protons that generate nearly mirror-image multiplets at 3.1 and 3.3?
Draw three-dimensional representation of the following amino acids:
(a) L-alanine
(b) L-Leucine
(c) D-serine
(d) D-glutamine
Show where trypsin and chymotrypsin would cleave the following peptide.
Tyr-Ile-Gln-Arg-Leu-Gly-Phe-Lys-Asn-Trp-Phe-Gly-Ala-Lys-Gly-Gln-Gln.NH2
Give equations for the formation and hydrogenolysis of the glutamine benzyl ester
After treatment with peroxyformic acid, the peptide hormone vasopressin is partially hydrolyzed. The following fragments are recovered. Propose a structure for vasopressin.
Phe-Gln-Asn Pro-Arg-Gly.NH2 Cys-Tyr-Phe
Asn-Cys-Pro-Arg Tyr-Phe-Gln-Asn
What do you think about this solution?
We value your feedback to improve our textbook solutions.