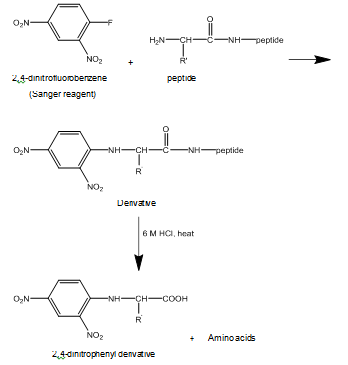
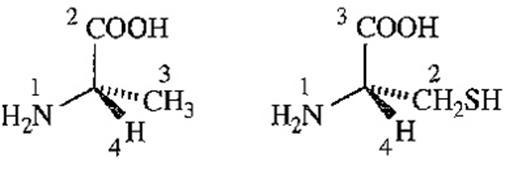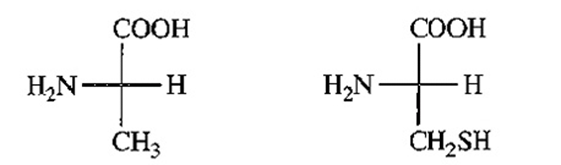Chapter 24: Q2P (page 1262)
Most naturally occurring amino acids have chiral centers (the asymmetric α carbon atoms) that are named (S) by the Cahn-Ingold-Prelog convention (Section 5-3). The common naturally occurring form of cysteine has a chiral center that is named (R), however.
(a) What is the relationship between (R)-Cysteine and (S)-alanine? Do they have the opposite three-dimensional configuration (as the names might suggest) or the same configuration?
(b) (S)-Alanine is an L-amino acid (Figure 24-2). Is (R)-cysteine a D-amino acid or an L-amino acid?
Short Answer
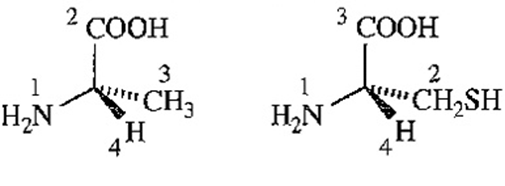
(S)-alanine (R)-cysteine
(a) The configurations around the asymmetric or chiral carbons of (R)-cysteine and (S)-alanine are same. The designation of configuration is different due to sulfur present in cysteine.
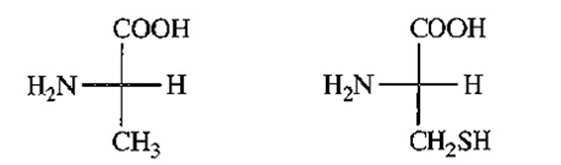
(S)-alanine (R)-cysteine
L-alanine L-cysteine
(b) Fischer projections of (S)-alanine and (R)-cysteine indicates that both are L-amino acids.