Chapter 24: Q1P (page 1262)
Draw three-dimensional representation of the following amino acids:
(a) L-alanine
(b) L-Leucine
(c) D-serine
(d) D-glutamine
Short Answer
(a), (b), (c) and (d) The diagrams are drawn below
Learning Materials
Features
Discover
Chapter 24: Q1P (page 1262)
Draw three-dimensional representation of the following amino acids:
(a) L-alanine
(b) L-Leucine
(c) D-serine
(d) D-glutamine
(a), (b), (c) and (d) The diagrams are drawn below
All the tools & learning materials you need for study success - in one app.
Get started for free
Show where trypsin and chymotrypsin would cleave the following peptide.
Tyr-Ile-Gln-Arg-Leu-Gly-Phe-Lys-Asn-Trp-Phe-Gly-Ala-Lys-Gly-Gln-Gln.NH2
Draw the electrophoretic separation of Trp, Cys, and His at pH 6.0.
Sometimes chemists need the unnatural D enantiomer of an amino acid, often as part of a drug or an insecticide. Most L-amino acids are isolated from proteins, but the D-amino acids are rarely found in natural proteins. D-amino acids can be synthesized from the corresponding L-amino acids. The following synthetic scheme is one of the possible methods.
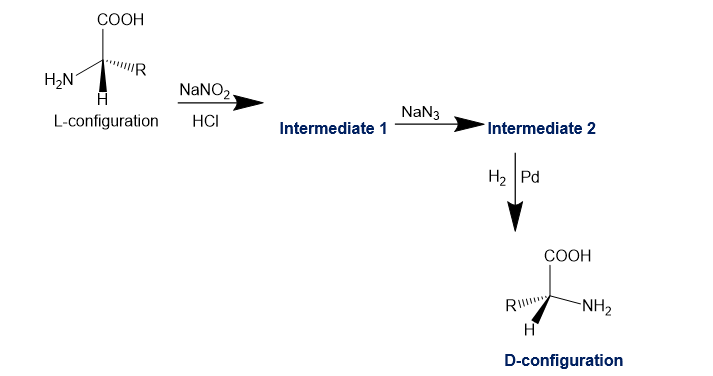
(a) Draw the structures of intermediates 1 and 2 in this scheme.
(b) How do we know that the product is entirely the unnatural D configuration?
Give equations for the formation and hydrogenolysis of the glutamine benzyl ester
What do you think about this solution?
We value your feedback to improve our textbook solutions.