Chapter 24: Q20P (page 1282)
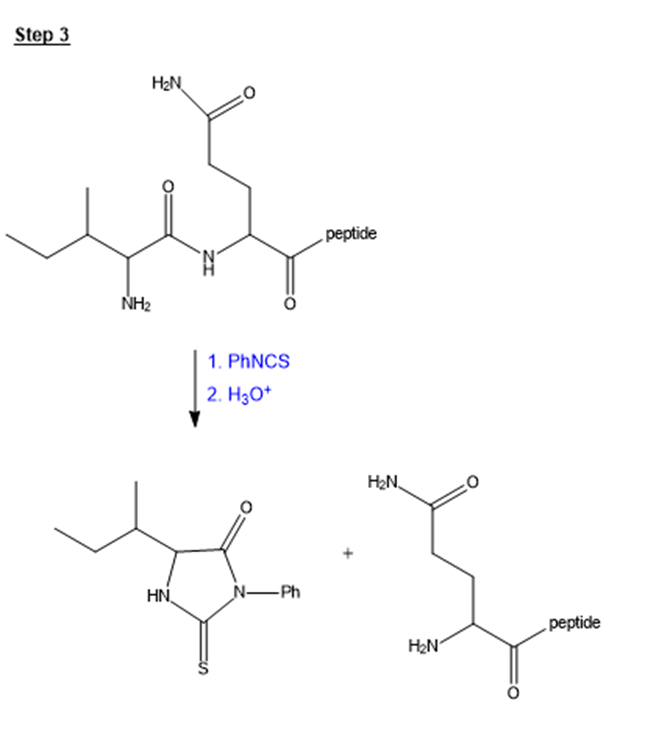
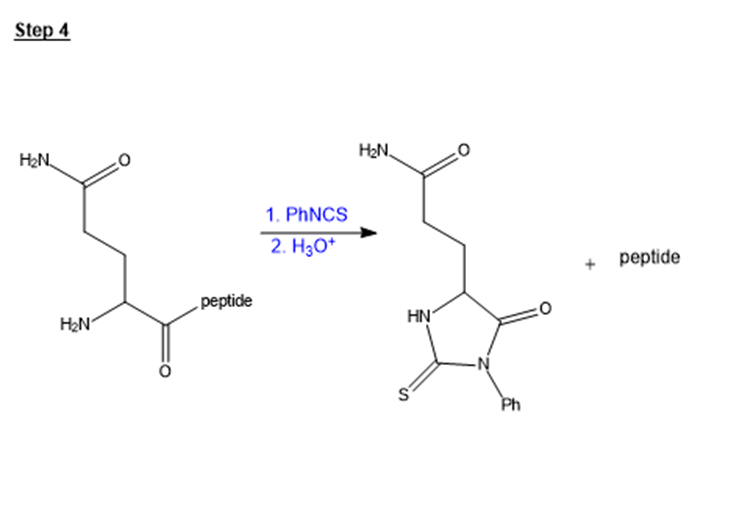
Show the third and fourth steps in the sequencing of oxytocin. Use figure 24-14 as a guide.
Short Answer
Learning Materials
Features
Discover
Chapter 24: Q20P (page 1282)
Show the third and fourth steps in the sequencing of oxytocin. Use figure 24-14 as a guide.


All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would use a Strecker synthesis to make
Answer
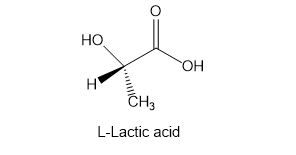
Suggest a method for the synthesis of the natural L enantiomer of alanine from the readily available L enantiomer of lactic acid.
The following structure is drawn in an unconventional manner.
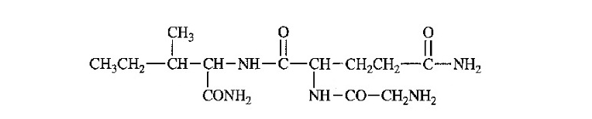
(a) Label the N terminus and the C terminus.
(b) Label the peptide bonds.
(c) Identify and label each amino acid present.
(d) Give the full name and the abbreviated name
Question: Propose a mechanism for the acid-catalyzed hydrolysis of phenylalanine ethyl ester.
Draw the resonance forms of a protonated guanidino group and explain why arginine has such a strongly basic isoelectric point.
What do you think about this solution?
We value your feedback to improve our textbook solutions.