Chapter 2: 37P (page 139)
Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do.
Learning Materials
Features
Discover
Chapter 2: 37P (page 139)
Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do.
All the tools & learning materials you need for study success - in one app.
Get started for free
Like nitrogen and carbon, oxygen also shows this same hybridization effect on acidity. Both of the following compounds can lose a proton from a positively charged oxygen with three bonds to give a conjugate base containing a neutral oxygen with two bonds. One of these structures has pKa = -2.4 , while the other has pKa = -8.0.
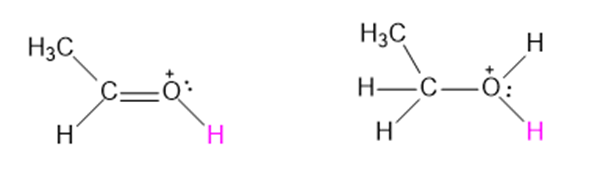
Compare the relative acidity of 1-molar aqueous solutions of the following acids.
Which is a stronger base: cyanide ion or fluoride ion? Give pKb values (without looking them up) to support your choice.
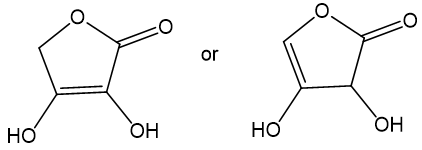
Choose the more acidic member of each pair of isomers, and show why the acid you chose is more acidic.
(a)
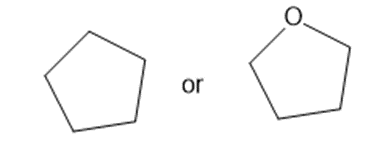
Predict which member of each pair is more soluble in water. Explain your prediction.
(a)
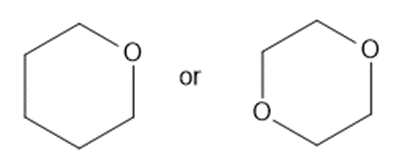
(b)
(c)
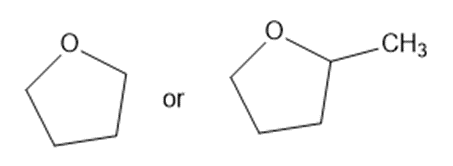
(d)
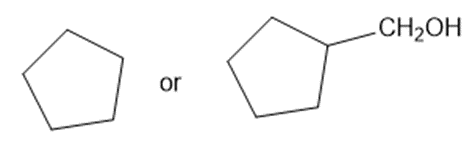
(e)
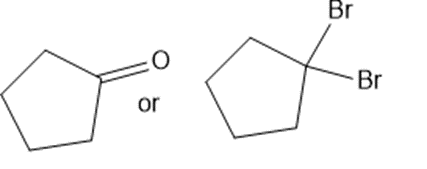
(f)
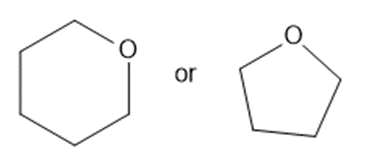
What do you think about this solution?
We value your feedback to improve our textbook solutions.