Chapter 2: 38P (page 139)
Rank the following species in order of increasing basicity. Explain your reasons for ordering them as you do.
Learning Materials
Features
Discover
Chapter 2: 38P (page 139)
Rank the following species in order of increasing basicity. Explain your reasons for ordering them as you do.
All the tools & learning materials you need for study success - in one app.
Get started for free
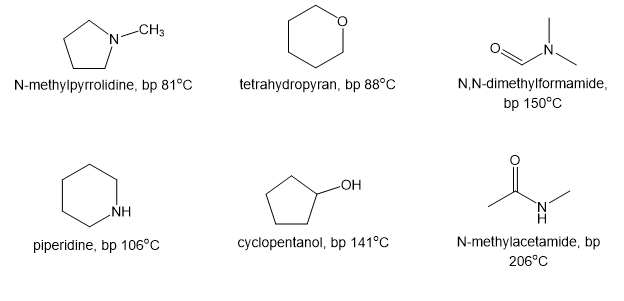
N-Methylpyrrolidine has a boiling point of, and piperidine has a boiling point of.
The following compounds can all react as acids.
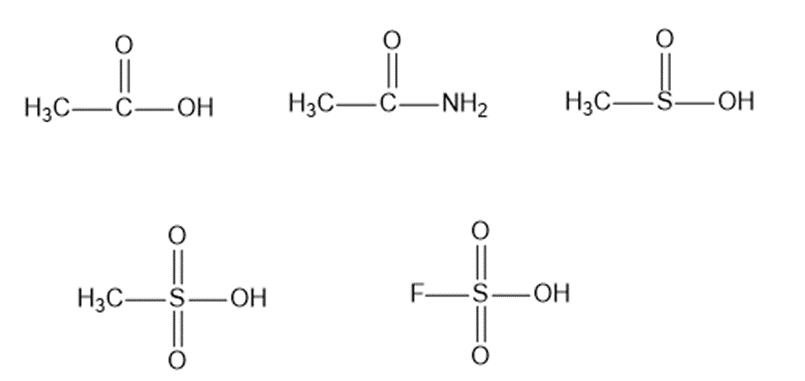
The of phenylacetic acid is, and theof propionic acid is.
(a)Calculate theof phenylacetic acid and theof propionic acid.
(b) Which one of these is the stronger acid? Calculate how much stronger an acid it is.
(c) Predict whether the following equilibrium will favor the reactants or the products.
Compare the relative acidity of 1-molar aqueous solutions of the following acids.
Predict which member of each pair is more soluble in water. Explain your prediction.
(a)
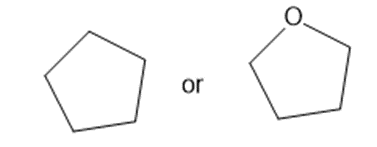
(b)
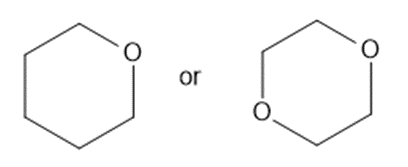
(c)
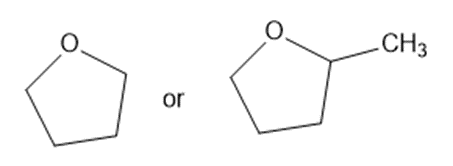
(d)
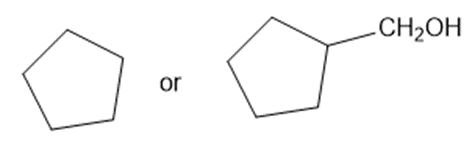
(e)
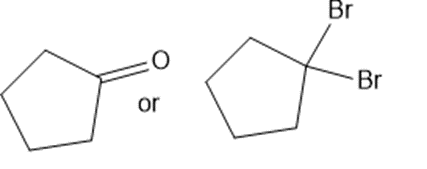
(f)
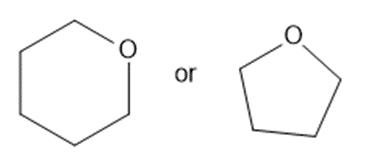
What do you think about this solution?
We value your feedback to improve our textbook solutions.