Chapter 2: Q32 P (page 138)
Predict which member of each pair is more soluble in water. Explain your prediction.
(a)
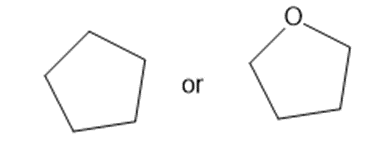
(b)
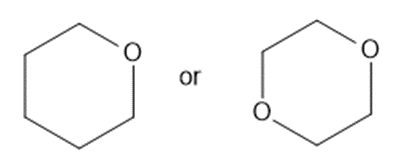
(c)
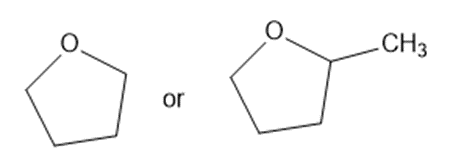
(d)
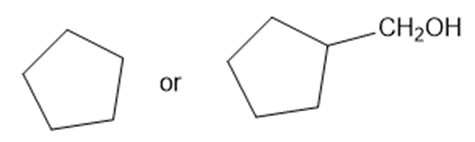
(e)
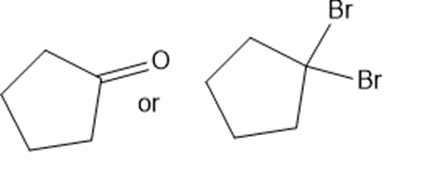
(f)
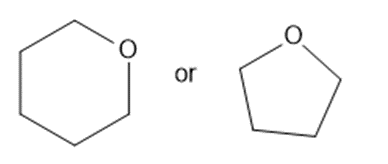
Short Answer
(a)
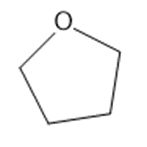
(b)
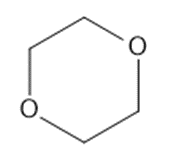
(c)
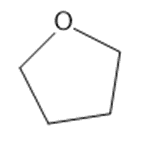
(d)
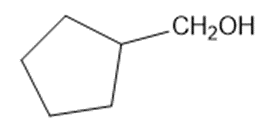
(e)
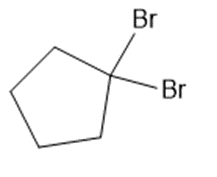
(f)
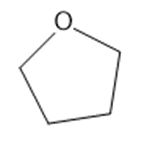
Learning Materials
Features
Discover
Chapter 2: Q32 P (page 138)
Predict which member of each pair is more soluble in water. Explain your prediction.
(a)

(b)

(c)

(d)

(e)

(f)

(a)

(b)

(c)

(d)

(e)

(f)

All the tools & learning materials you need for study success - in one app.
Get started for free
The following compound can become protonated on any of the three nitrogen atoms. One of these nitrogens is much more basic than the others, however.
The C ≡ N triple bond in acetonitrile has a dipole moment of about 3.6Dand a bond length of about 1.16Å. Calculate the amount of charge separation in this bond. How important is the charge separated resonance form in the structure of acetonitrile?

Predict the products of the following acid-base reactions.
(a)
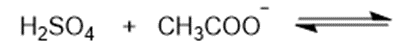
(b)
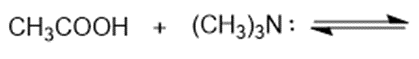
(c)
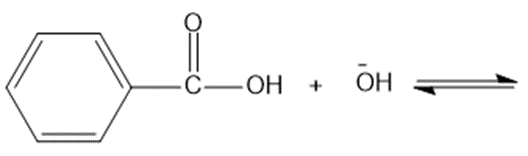
(d)
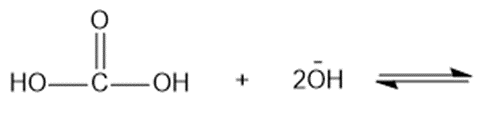
(e)

(f)
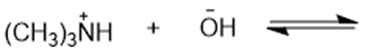
(g)
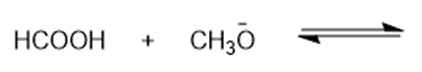
(h)
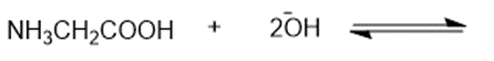
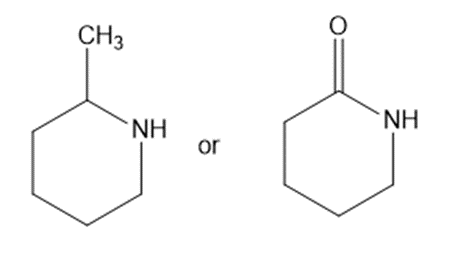
Predict which compound in each pair has the higher boiling point. Explain your prediction.
(a)
CH3CH2CH2OCH3 or CH3CH2CH2(OH)CH3
(b)
CH3CH2CH2CH2CH3 or CH3CH2CH2CH2CH2CH3
(c)
CH3CH2CH2CH2CH2CH3 or (CH2)3CHCH2CH2CH3
(d)
CH3CH2CH2CH2CH2CH3 or CH3CH2CH2CH2CH2CH2Cl
(e)
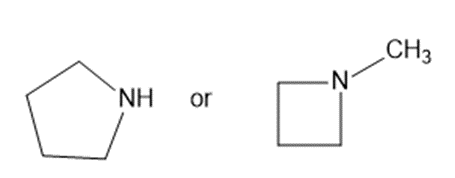
(f)
TheC=Odouble bond has a dipole moment of about 2.4Dand a bond length of about 1.23 Å.
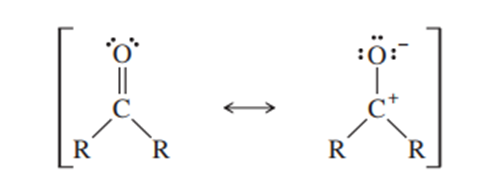
(R is a general symbol for a carbon-containing group)
What do you think about this solution?
We value your feedback to improve our textbook solutions.