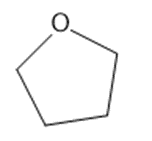
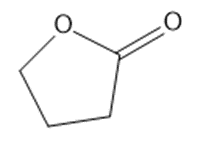
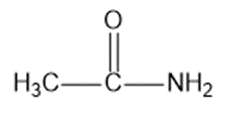
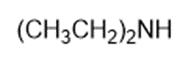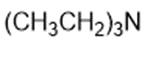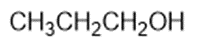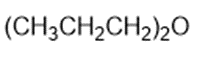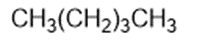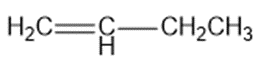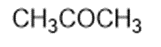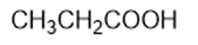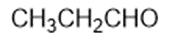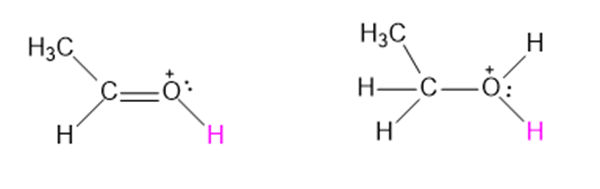Chapter 2: Q1P (page 93)
TheC=Odouble bond has a dipole moment of about 2.4Dand a bond length of about 1.23 Å.
- Calculate the amount of charge separation in this bond.
- Use this information to evaluate the relative importance of the following two resonance contributors:
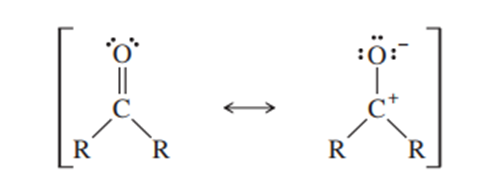
(R is a general symbol for a carbon-containing group)
Short Answer
- Amount of charge separation is δ = 0.4065 e.
- There is 40% of the second resonance structure and 60% of the first resonance structure in resonance equilibrium.