Chapter 2: Q2P (page 95)
The N-F bond is more polar than N-H bond, but NF3 has smaller dipole moment than NH3. Explain the curious result.
NH3: µ= 1.5 D, NF3: µ= 0.2D
Short Answer
N—F bond is more polar than N—H but, NF3 is less polar than NH3 because in NF3 individual polarity face towards F therefore net will cancel (since it is very less). But in NH3 N is more electronegative than H therefore this all add up and net is more.



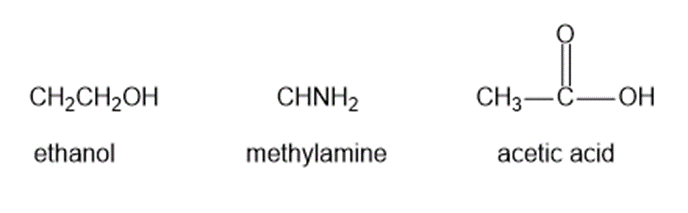
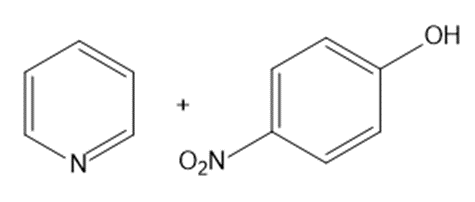
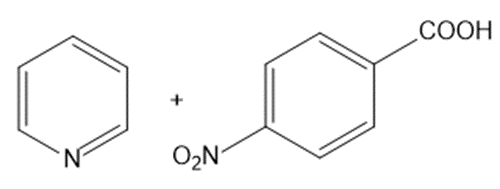
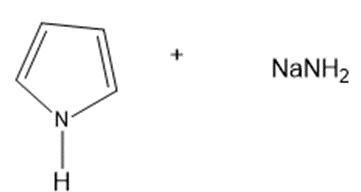
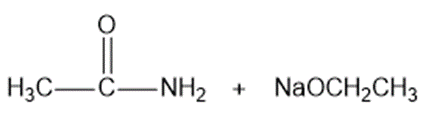
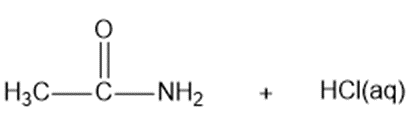
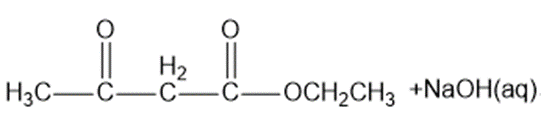
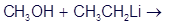
 ? Would you expect
? Would you expect to be a strong base or a weak base?
to be a strong base or a weak base?