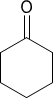
Chapter 2: Q 49P (page 140)
Question: Ethyllithium is often used as a base in organic reactions,
(a)Predict the products of the following acid-base reaction,
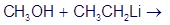
(b) What is the conjugate acid of ? Would you expect
? Would you expect to be a strong base or a weak base?
to be a strong base or a weak base?
Short Answer
(a)
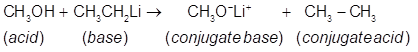
(b)
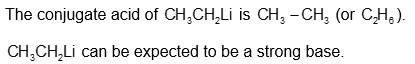
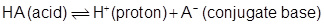
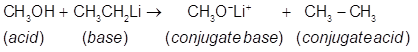


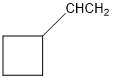
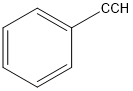
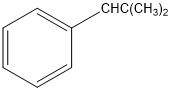
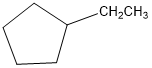
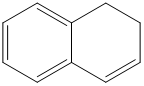
 or
or