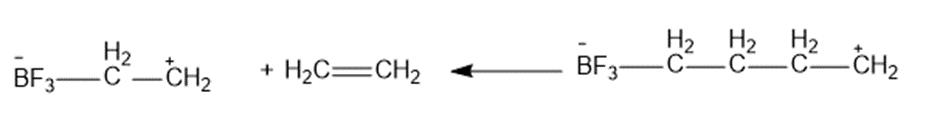
Chapter 2: Q28P (page 137)
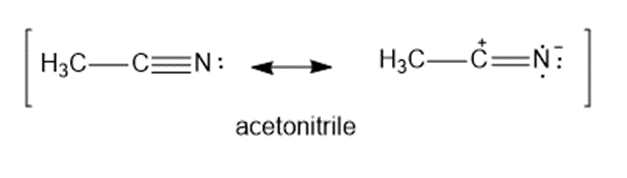
The C ≡ N triple bond in acetonitrile has a dipole moment of about 3.6Dand a bond length of about 1.16Å. Calculate the amount of charge separation in this bond. How important is the charge separated resonance form in the structure of acetonitrile?
Short Answer
Amount of charge separation is δ=0.6465e .
There is 64.65% of the charge separated resonance structure in resonance equilibrium.


 or
or