Chapter 2: Q42 P (page 139)
Predict the products of the following acid-base reactions.
(a)
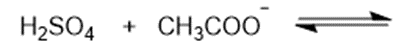
(b)
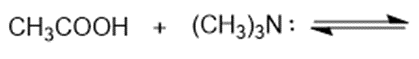
(c)
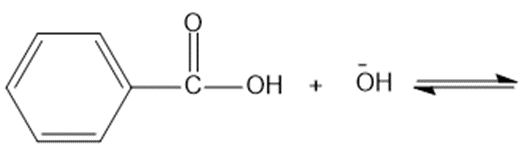
(d)
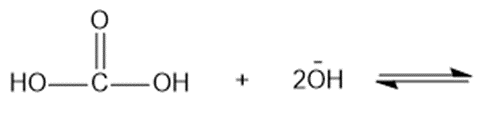
(e)

(f)
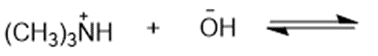
(g)
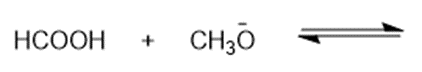
(h)
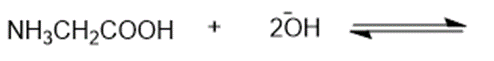
Short Answer
(a)
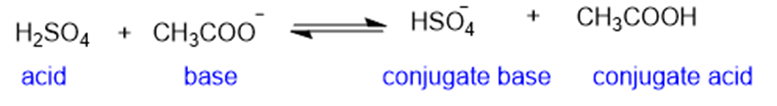
(b)
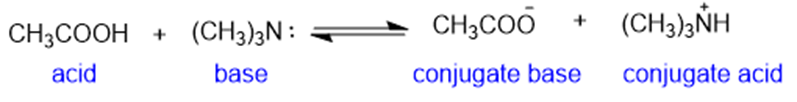
(c)
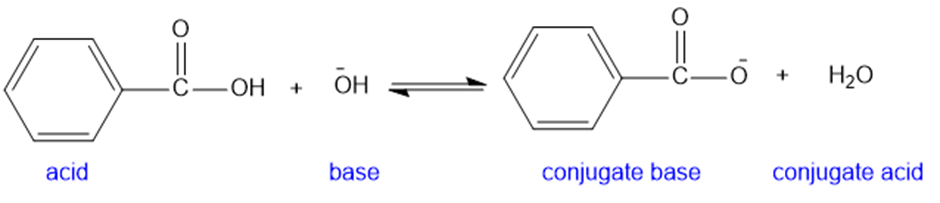
(d)
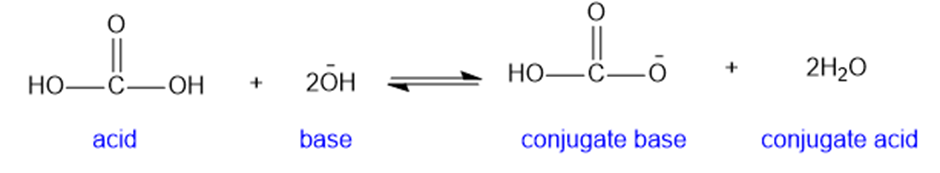
(e)
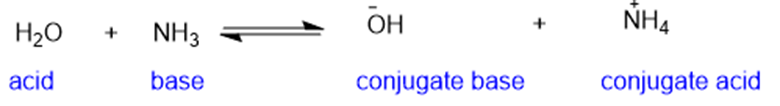
(f)

(g)
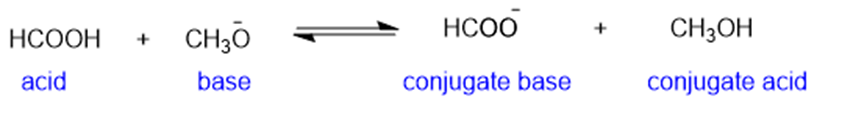
(h)
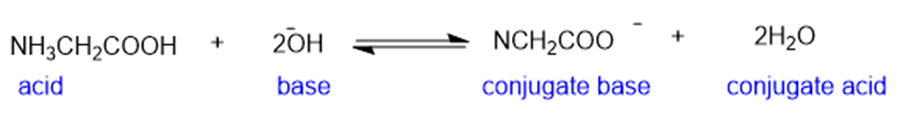
Learning Materials
Features
Discover
Chapter 2: Q42 P (page 139)
Predict the products of the following acid-base reactions.
(a)
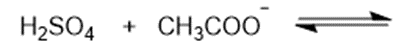
(b)
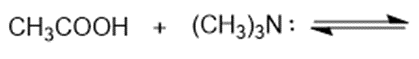
(c)

(d)
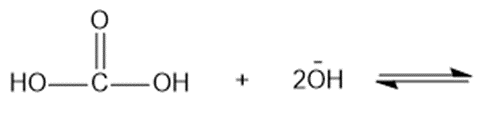
(e)

(f)
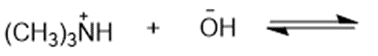
(g)
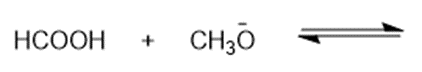
(h)
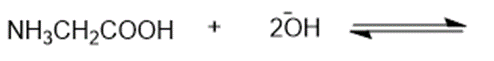
(a)

(b)

(c)

(d)

(e)
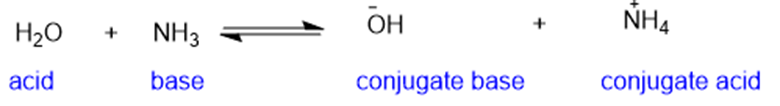
(f)

(g)

(h)

All the tools & learning materials you need for study success - in one app.
Get started for free
Ethyllithium is often used as a base in organic reactions,
(a)Predict the products of the following acid-base reaction,
CH3OH + CH3CH2Li
(b) What is the conjugate acid ofCH3CH2Li ? Would you expectCH3CH2Li to be a strong base or a weak base?
Question: Circle the functional groups in the following structures. State to which class (or classes) of compounds the structure belongs.
(a)
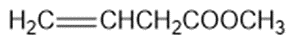
(b)
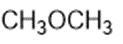
(c)
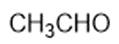
(d)

(e)

(f)
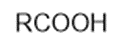
(g)
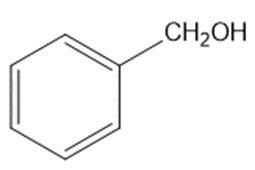
(h)
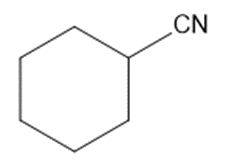
(i)
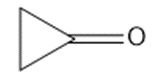
(j)
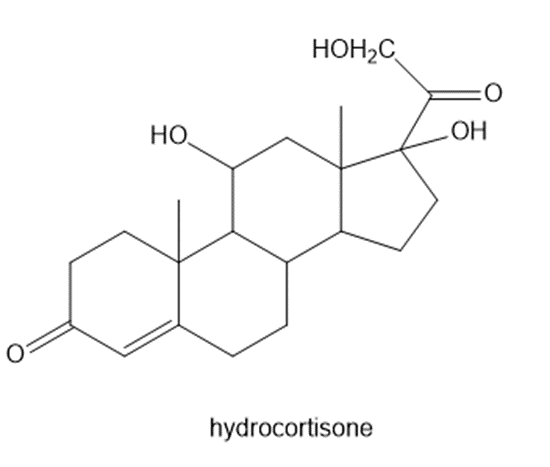
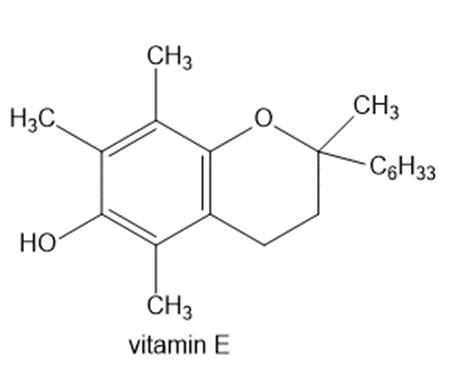
(k)
Ozone has a dipole moment of 0.53 D. Carbon dioxide has a dipole moment of zero, even though C-O bonds are more polar than O-O bonds. Explain this apparent contradiction.
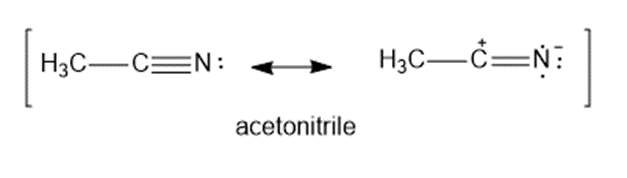
Question: The C ≡ Ntriple bond in acetonitrile has a dipole moment of about 3.6 Dand a bond length of about 1.16 Å. Calculate the amount of charge separation in this bond. How important is the charge separated resonance form in the structure of acetonitrile?
The following compounds can all react as bases.
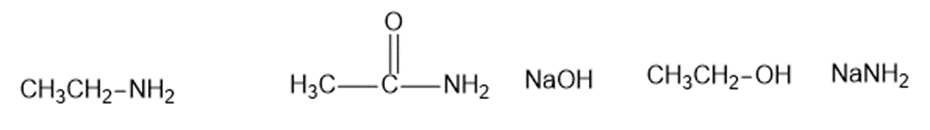
What do you think about this solution?
We value your feedback to improve our textbook solutions.