Chapter 2: Q21P (page 121)
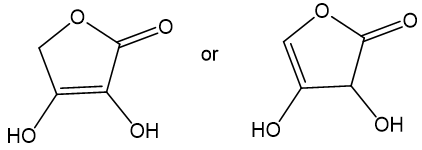
Choose the more acidic member of each pair of isomers, and show why the acid you chose is more acidic.
(a)
Short Answer
The more acidic among the given pair is
Learning Materials
Features
Discover
Chapter 2: Q21P (page 121)
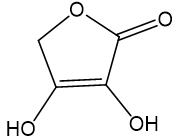
Choose the more acidic member of each pair of isomers, and show why the acid you chose is more acidic.
(a)

The more acidic among the given pair is

All the tools & learning materials you need for study success - in one app.
Get started for free
Classify the following hydrocarbons, and draw a lewis structure for each one. A compound may fit into more than one of the following classifications:
Alkane, alkene, alkyne, cycloalkane, cycloalkene, cycloalkyne, aromatic hydrocarbon.
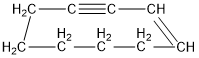
e.
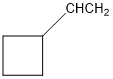
f.
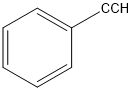
g.
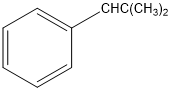
h.
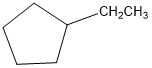
i.
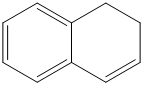
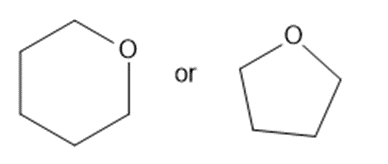
Predict which member of each pair is more soluble in water. Explain your prediction.
(a)
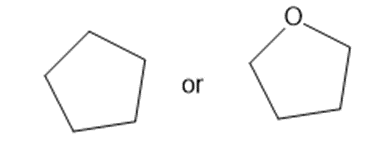
(b)
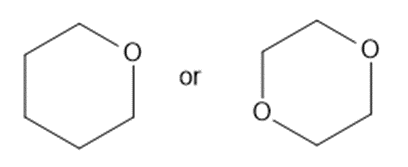
(c)
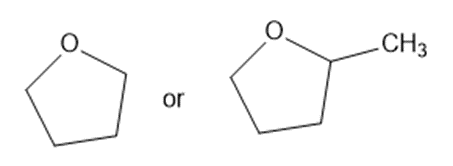
(d)
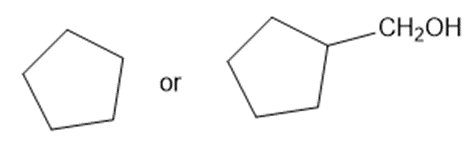
(e)
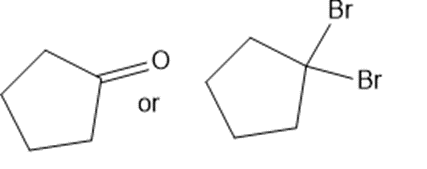
(f)
Draw a Lewis structure, and classify each of the following compounds.
(a)
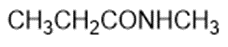
(b)
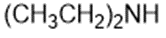
(c)

(d)

(e)
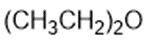
(f)
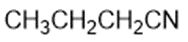
(g)
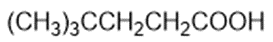
(h)
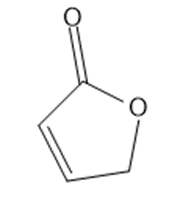
(i)
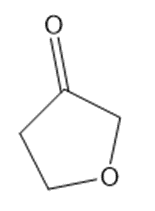
(j)
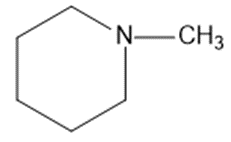
(k)
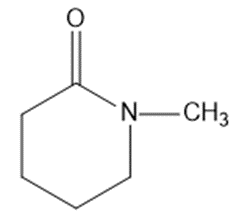
(l)
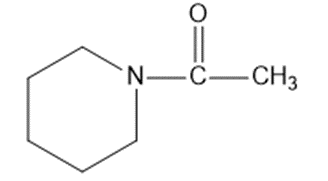
(m)
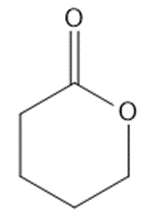
(n)
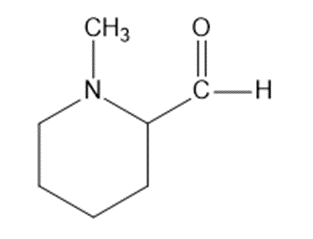
(o)
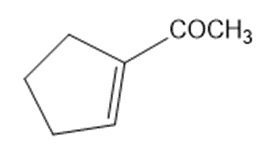
Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do.
Compare the relative acidity of 1-molar aqueous solutions of the following acids.
What do you think about this solution?
We value your feedback to improve our textbook solutions.