Chapter 2: Q18P (page 119)
Which is a stronger base: cyanide ion or fluoride ion? Give pKb values (without looking them up) to support your choice.
Short Answer
The stronger base is the cyanide ion.
pKb(CN-) = 4.69
pKb(F-) = 10.80
Learning Materials
Features
Discover
Chapter 2: Q18P (page 119)
Which is a stronger base: cyanide ion or fluoride ion? Give pKb values (without looking them up) to support your choice.
The stronger base is the cyanide ion.
pKb(CN-) = 4.69
pKb(F-) = 10.80
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Ethyllithium is often used as a base in organic reactions,
(a)Predict the products of the following acid-base reaction,
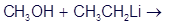
(b) What is the conjugate acid of ? Would you expect
? Would you expect to be a strong base or a weak base?
to be a strong base or a weak base?
The of phenylacetic acid is, and theof propionic acid is.
(a)Calculate theof phenylacetic acid and theof propionic acid.
(b) Which one of these is the stronger acid? Calculate how much stronger an acid it is.
(c) Predict whether the following equilibrium will favor the reactants or the products.
Calculate the pH of the following solutions:
(a) 5.00 g of HBr in 100mL of aqueous solution
(b) 1.50 g of NaOH in 50mL of aqueous solution
Two isomers of 1,2 Dichloroethene are known. One has dipole moment of 2.4 D; the other has zero dipole moment. Draw the two isomers, and explain why one has zero dipole moment
CHCl=CHCl (1,2 Dichloroethene)
Consider the following compounds that vary from nearly nonacidic to strongly acidic. Draw the conjugate bases of these compounds and explain why the acidity increases so dramatically with substitution by nitro groups.
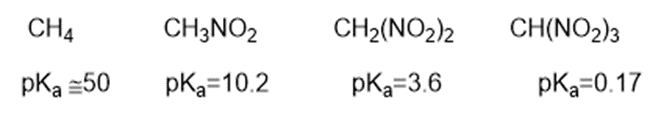
What do you think about this solution?
We value your feedback to improve our textbook solutions.