Chapter 2: Q17P (page 119)
Consider each pair of bases, and explain which one is more basic. Draw their conjugate acids, and show which one is a stronger acid.
(a)
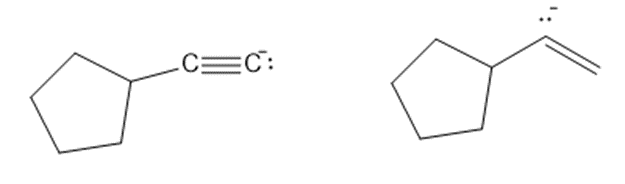
(b)
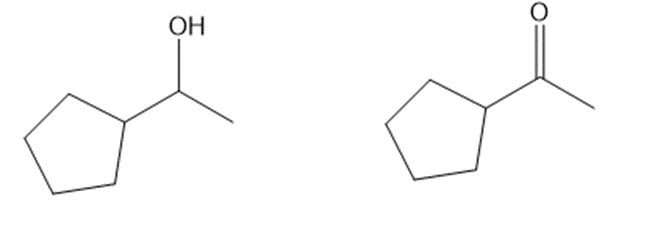
(c)
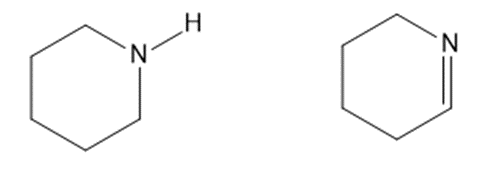
(d)
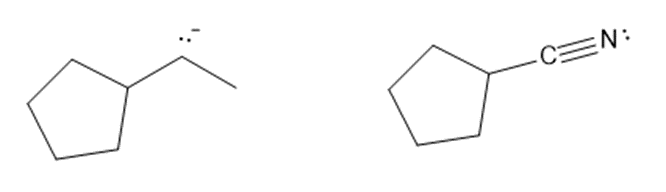
Short Answer
(a)
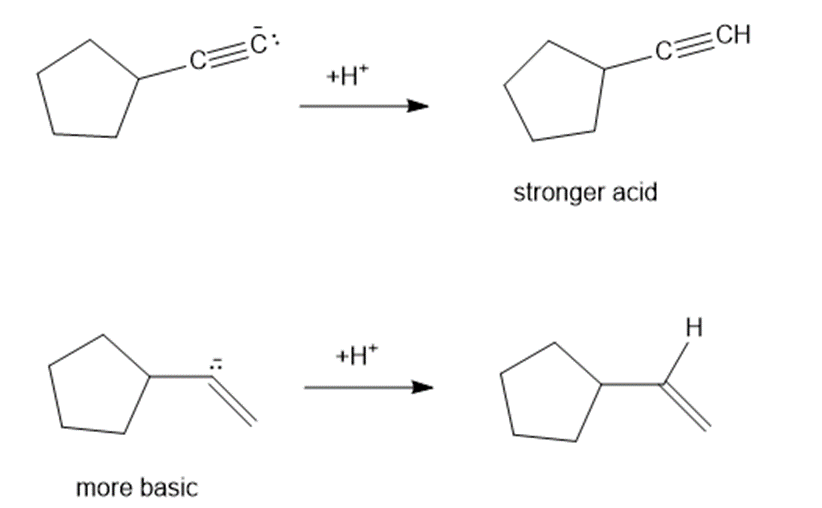
(b)
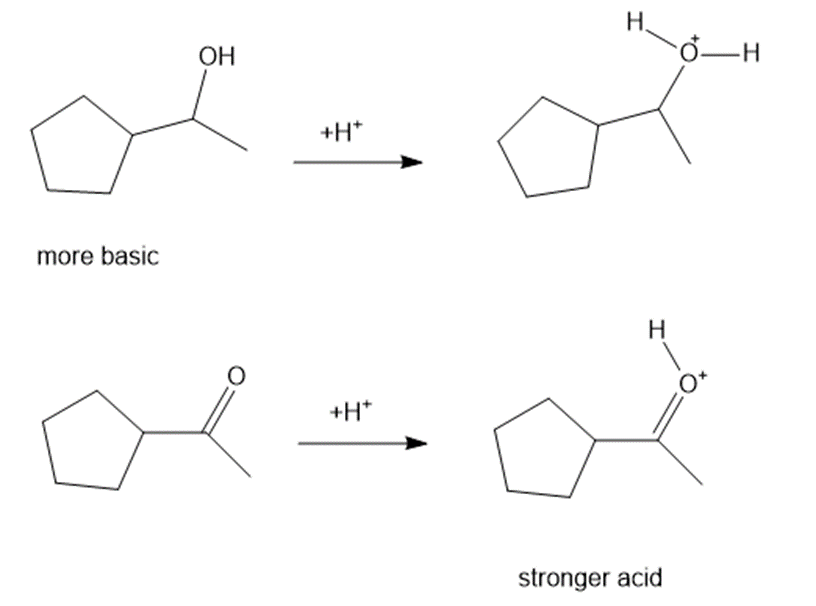
(c)
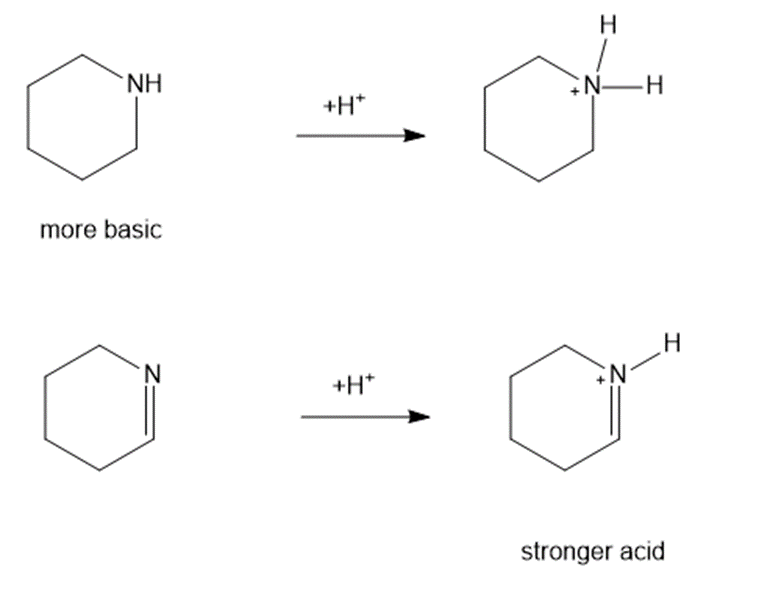
(d)
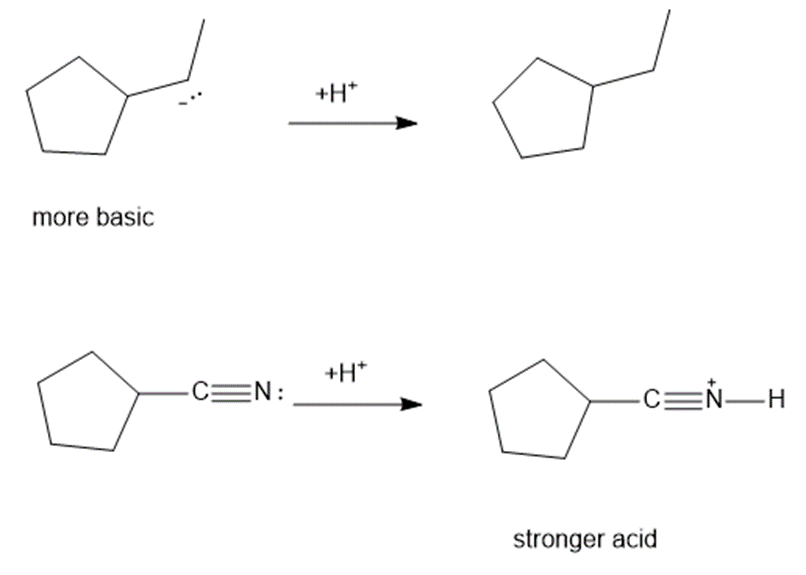
Learning Materials
Features
Discover
Chapter 2: Q17P (page 119)
Consider each pair of bases, and explain which one is more basic. Draw their conjugate acids, and show which one is a stronger acid.
(a)

(b)

(c)

(d)

(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
Circle the functional groups in the following structures. State to which class (or classes) of compounds the structure belongs.
(a)
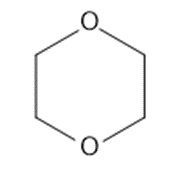
(b)
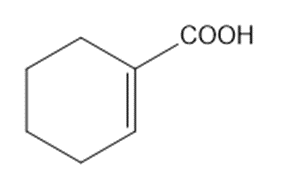
(c)
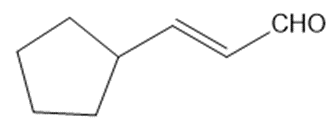
(d)
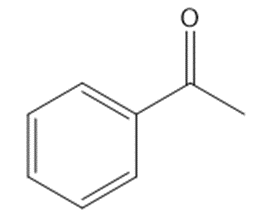
(e)
(f)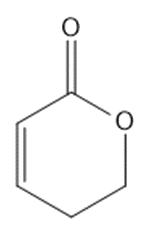
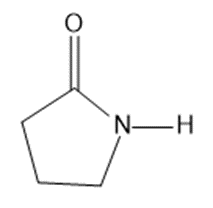
(g)
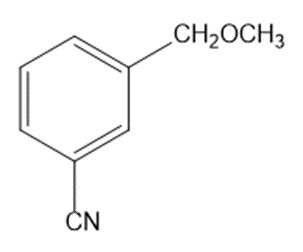
(h)
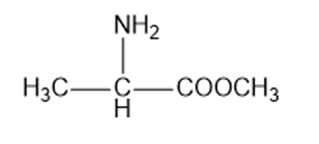
(i)
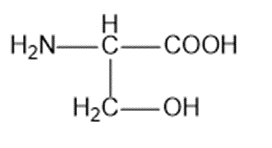
Calculate the pH of the following solutions:
(a) 5.00 g of HBr in 100mL of aqueous solution
(b) 1.50 g of NaOH in 50mL of aqueous solution
Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do.
Consider the type of orbitals involved, and rank the following nitrogen compounds in order of decreasing basicity. Give the structure of their conjugate acids; and estimate their pKas from similar compounds in Appendix 4. Rank the conjugate acids in order of increasing acidity. (Hint: These two orders should be the same!)
Circle the member of each pair that is more soluble in water.
(a) CH3CH2OCH2CH3 or CH3CH2CH2CH2CH3
(b) CH3CH2OCH2CH3 or CH3CH2CH2OH
(c) CH3CH2NHCH3 or CH3CH2CH2CH3
(d) CH3CH2OH or CH3CH2CH2CH2OH
(e)  or
or 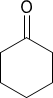
What do you think about this solution?
We value your feedback to improve our textbook solutions.