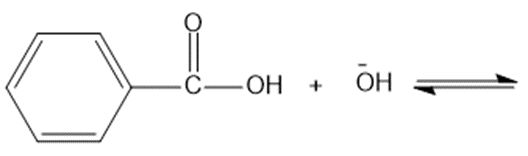
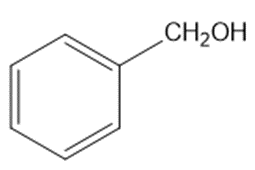
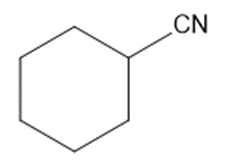
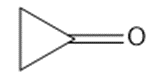
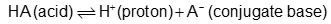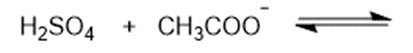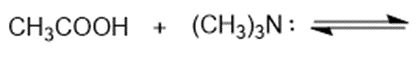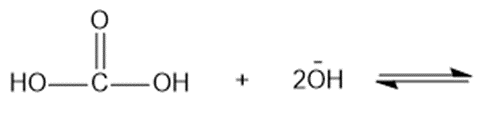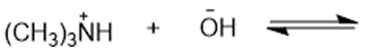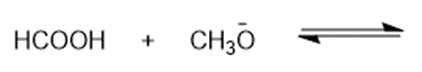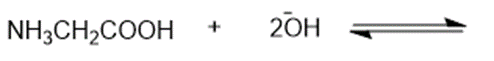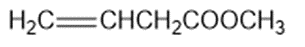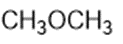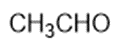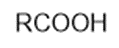Chapter 2: Q48 P (page 140)
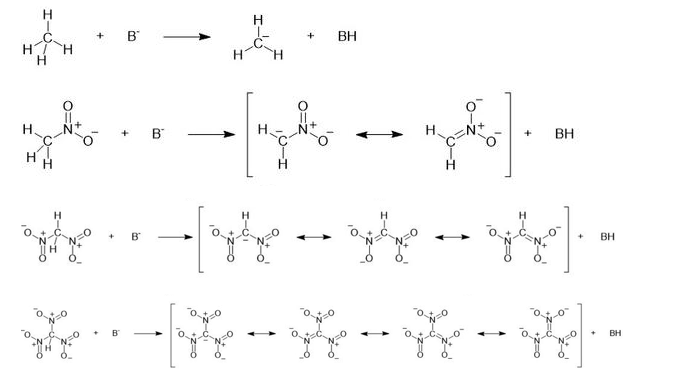
Consider the following compounds that vary from nearly nonacidic to strongly acidic. Draw the conjugate bases of these compounds and explain why the acidity increases so dramatically with substitution by nitro groups.
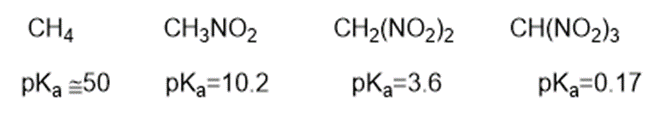
Short Answer
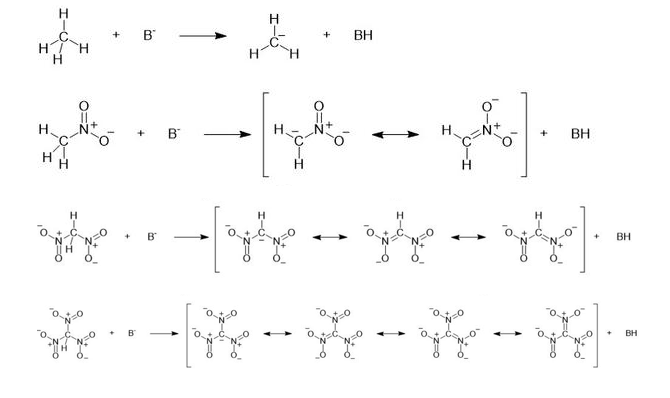
The anions (conjugate bases) are stabilized by delocalization.