Chapter 30: Question 30.46 (page 1227)
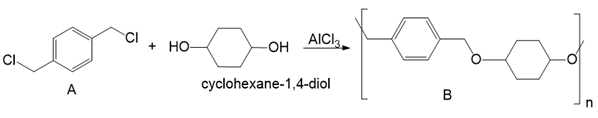
Draw a stepwise mechanism for the conversion of dihalide A and cyclohexane-1,4-diol to polyether B in the presence of AlCl3.
Short Answer
Answer
 Mechanism of the reaction
Mechanism of the reaction
Learning Materials
Features
Discover
Chapter 30: Question 30.46 (page 1227)
Draw a stepwise mechanism for the conversion of dihalide A and cyclohexane-1,4-diol to polyether B in the presence of AlCl3.

Answer
 Mechanism of the reaction
Mechanism of the reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
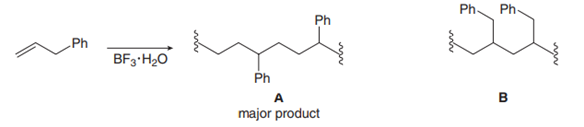
Cationic polymerization of 3-phenylpropene ()affords A as the major product rather than B. Draw a stepwise mechanism to account for this observation.
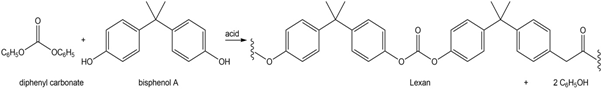
Lexan can also be prepared by the acid-catalyzed reaction of diphenyl carbonate with bisphenol A. Draw a stepwise mechanism for this process.
Why can’t chemical recycling—that is, the conversion of polymer to monomers and re-conversion of monomers to polymer—be done easily with HDPE and LDPE?
What monomer(s) are used to prepare each polymer or copolymer?
(a.)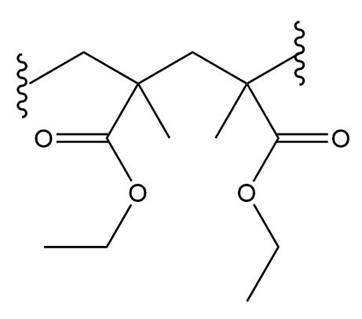
(b.)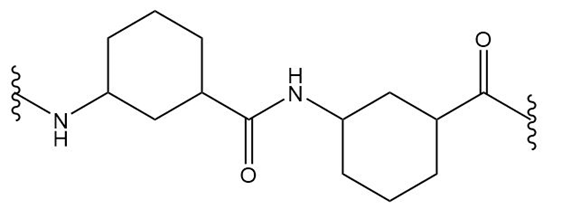
(c.)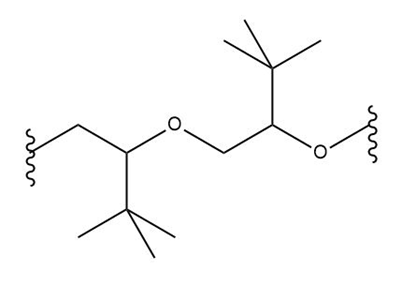
(d.)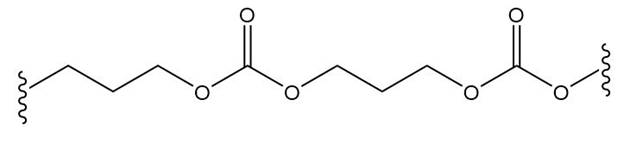
Draw a stepwise mechanism for the conversion of acrylonitrile to polyacrylonitrile, , using butyllithium(BuLi) as the initiator and CO2 as the electrophile to terminate the chain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.