Chapter 28: Question 28.54 (page 1149)
What products are formed when each compound is subjected to a Kiliani–Fischer synthesis?
(a.)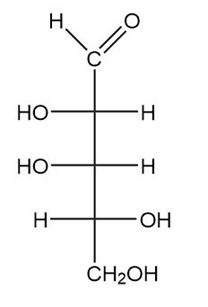
(b.)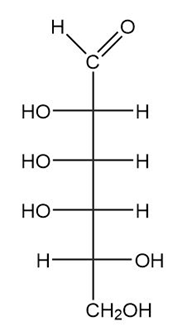
Short Answer
Answer
(a.)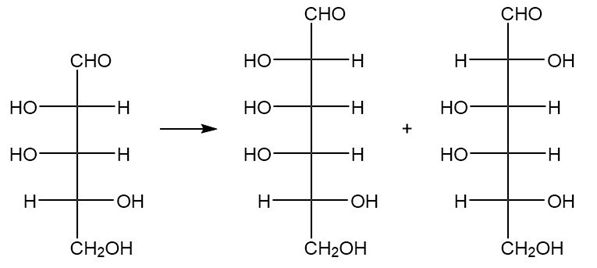
(b.)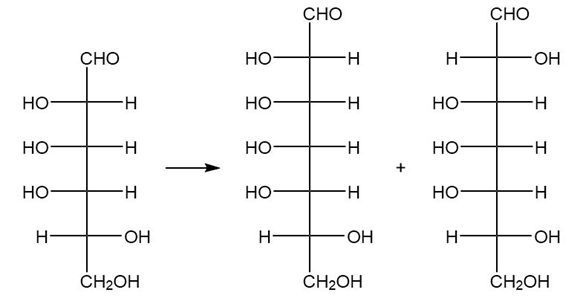
Learning Materials
Features
Discover
Chapter 28: Question 28.54 (page 1149)
What products are formed when each compound is subjected to a Kiliani–Fischer synthesis?
(a.)
(b.)
Answer
(a.)
(b.)
All the tools & learning materials you need for study success - in one app.
Get started for free
Which aldoses are oxidized to optically inactive aldaric acids: (a) D-erythrose; (b) D-lyxose; (c) D-galactose?
Convert each compound to a Fischer projection and label each stereogenic center as R or S.
(a.)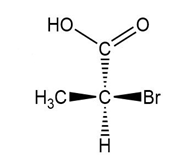
(b.)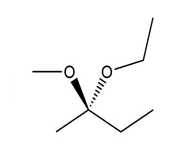
(c.)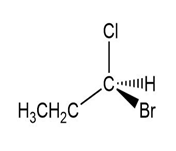
(d.)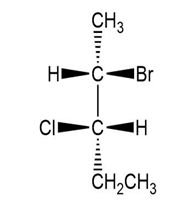
(e.)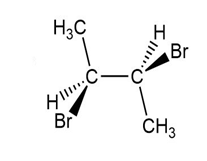
(f.)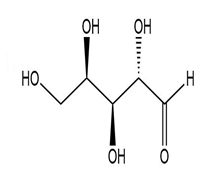
(a) Why can’t two purine bases (A and G) form a base pair and hydrogen bond to each other on two strands of DNA in the double helix? (b) Why is hydrogen bonding between guanine and cytosine more favorable than hydrogen bonding between guanine and thymine?
Draw the structures of the nucleosides formed from each of the following components: (a) ribose + uracil; (b) 2-deoxyribose + guanine.
(a) Convert each cyclic monosaccharide into a Fischer projection of its acyclic form. (b) Name each monosaccharide. (c) Label the anomer as α or β.
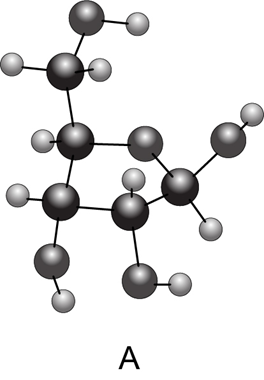
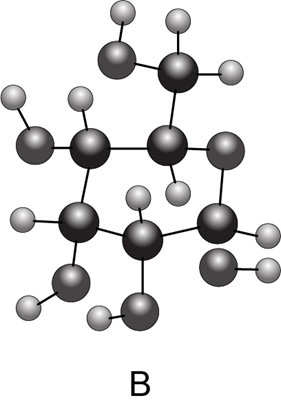
What do you think about this solution?
We value your feedback to improve our textbook solutions.