Chapter 28: Question 28.53 (page 1149)
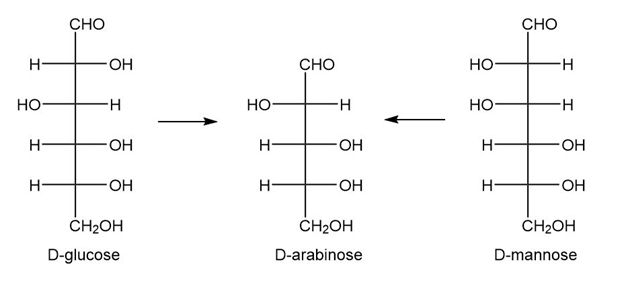
What two aldohexoses yield d-arabinose upon Wohl degradation?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 28: Question 28.53 (page 1149)
What two aldohexoses yield d-arabinose upon Wohl degradation?
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Treating chitin with hydrolyzes its amide linkages, forming a compound called chitosan. What is the structure of chitosan? Chitosan has been used in shampoos, fibers for sutures, and wound dressings.
Draw a Fischer projection of the monosaccharide from which each of the following glycosides was prepared.
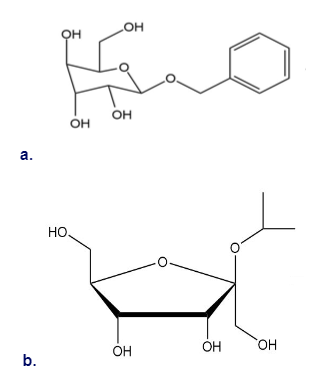
Draw the products formed when is treated with each reagent.
a.
b.
c. , pyridine
d. The product in (a), then
e. The product in (b), then , pyridine
f. The product in (d), then
Draw a stepwise mechanism for the following reaction.
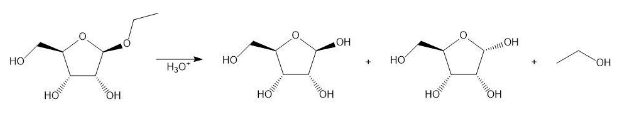
D-Arabinose can exist in both pyranose and furanose forms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.