Chapter 28: Question 28.18 (page 1125)
Draw a stepwise mechanism for the following reaction.
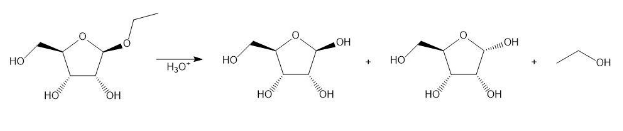
Short Answer
Answer
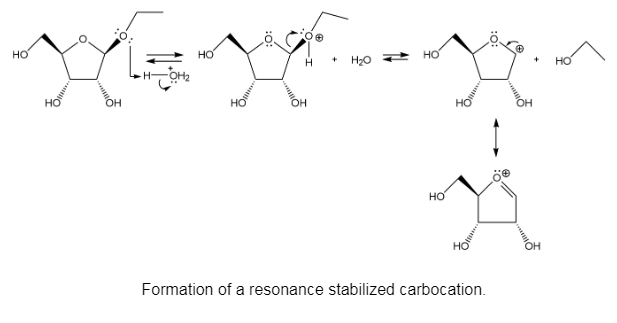
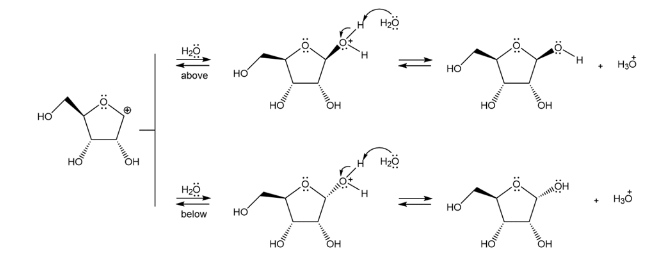
The mechanism of the above reaction is followed by resonance and nucleophilic attack of H2O to form anomers.
Learning Materials
Features
Discover
Chapter 28: Question 28.18 (page 1125)
Draw a stepwise mechanism for the following reaction.

Answer
The mechanism of the above reaction is followed by resonance and nucleophilic attack of H2O to form anomers.


All the tools & learning materials you need for study success - in one app.
Get started for free
Which d-aldopentoses are reduced to optically inactive alditols using ?
How would you convert d-glucose into each compound? More than one step is required.
(a.)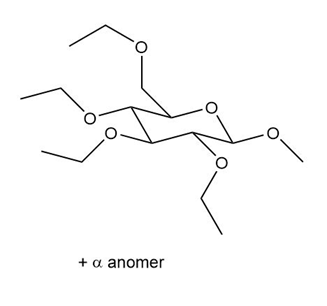
(b.)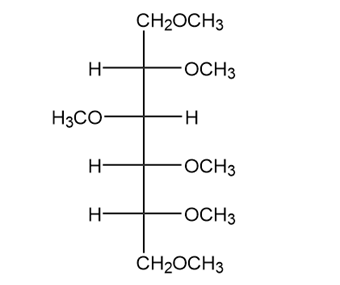
(c.)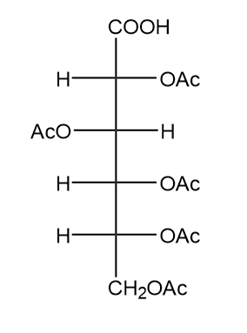
Drawthe structure of:
(a) a polysaccharide formed by joining d-mannose units in -glycosidic linkages;
(b) a polysaccharide formed by joining d-glucose units in -glycosidic linkages.
The polysaccharide in (b) is dextran, a component of dental plaque.
Consider the tetrasaccharide stachyose drawn below. Stachyose is found in white jasmine, soybeans, and lentils. Because humans cannot digest it, its consumption causes flatulence.
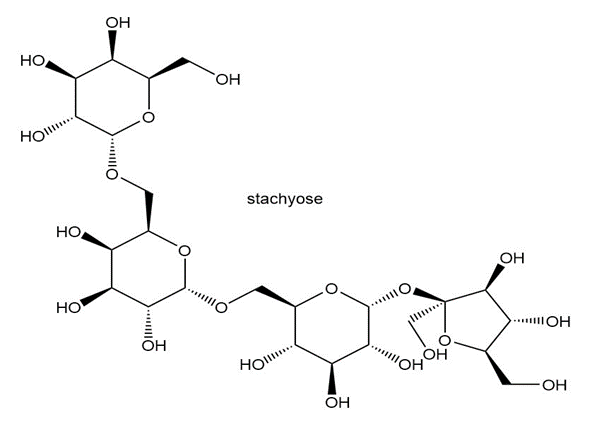
a. Label all glycoside bonds.

c. What products are formed when stachyose is hydrolyzed with H3O+?
d. Is stachyose a reducing sugar?
e. What product is formed when stachyose is treated with excess CH3I, Ag2O?
f. What products are formed when the product in (e) is treated with H3O+?
Draw the products formed when D-arabinose is treated with each reagent. (a) ; (b) ; (c) ,
What do you think about this solution?
We value your feedback to improve our textbook solutions.