Chapter 28: Question 28.40 (page 1147)
Convert each compound to a Fischer projection and label each stereogenic center as R or S.
(a.)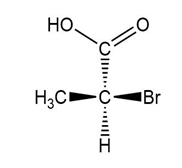
(b.)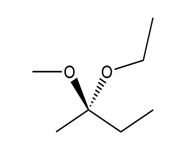
(c.)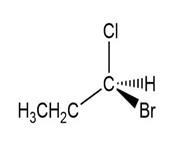
(d.)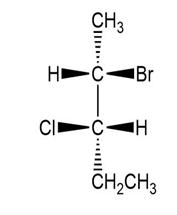
(e.)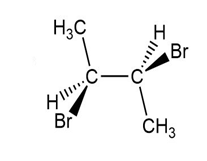
(f.)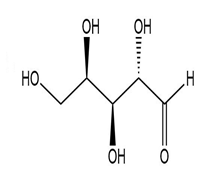
Short Answer
Answer
(a.)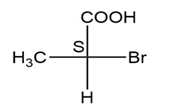
(b.)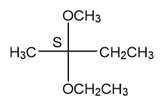
(c.)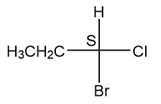
(d.)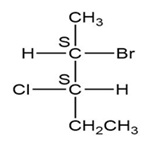
(e.)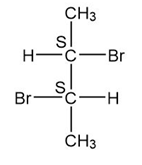
(f.)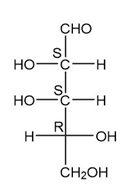
Learning Materials
Features
Discover
Chapter 28: Question 28.40 (page 1147)
Convert each compound to a Fischer projection and label each stereogenic center as R or S.
(a.)
(b.)
(c.)
(d.)
(e.)
(f.)
Answer
(a.)
(b.)
(c.)
(d.)
(e.)
(f.)
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a Haworth projection for each compound using the structures in Figures 28.4 and 28.5.

Convert each cyclic monosaccharide into its acyclic form.
(a.)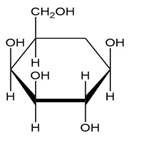
(b.)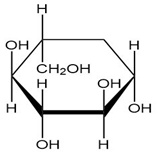
(c.)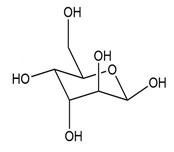
(d.)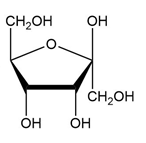
a. Identify the glycosidic linkage in disaccharide C, classify the glycosidic bond as , and use numbers to designate its location.
b. Identify the lettered compounds in the following reaction.
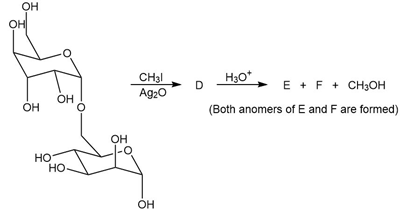
D-Arabinose can exist in both pyranose and furanose forms.
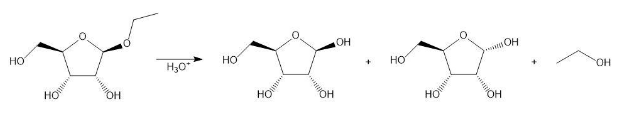
Draw a stepwise mechanism for the following reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.