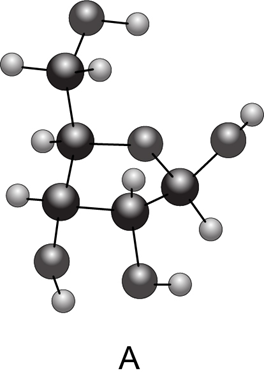
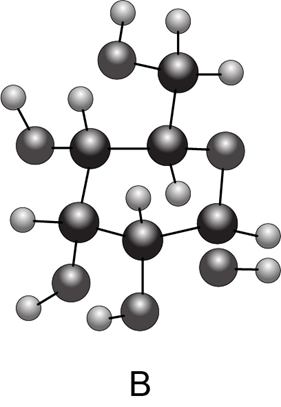
Chapter 28: Question 28.38 (page 1147)
(a) Convert each cyclic monosaccharide into a Fischer projection of its acyclic form. (b) Name each monosaccharide. (c) Label the anomer as α or β.
Short Answer
Answer
 Compound A
Compound A
 Compound B
Compound B
Learning Materials
Features
Discover
Chapter 28: Question 28.38 (page 1147)
(a) Convert each cyclic monosaccharide into a Fischer projection of its acyclic form. (b) Name each monosaccharide. (c) Label the anomer as α or β.


Answer
 Compound A
Compound A
 Compound B
Compound B
All the tools & learning materials you need for study success - in one app.
Get started for free
Which aldoses are oxidized to optically inactive aldaric acids: (a) D-erythrose; (b) D-lyxose; (c) D-galactose?
Which D-aldopentose is oxidized to an optically active aldaric acid and undergoes the Wolf degradation to yield a D-aldotetrose that is oxidized to an optically active aldaric acid?
Convert each cyclic monosaccharide into its acyclic form.
(a.)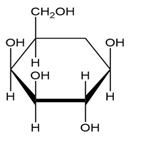
(b.)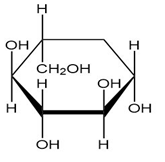
(c.)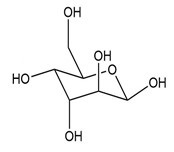
(d.)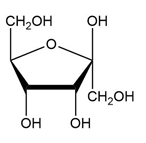
Draw a stepwise mechanism for the conversion of β-D-glucose to both anomers of N-ethyl glucopyranoside, the equation written in Reaction [1].
Draw both pyranose anomers of each aldohexose using a three-dimensional representation with a chair pyranose. Label each anomers as .
(a.)
(b.)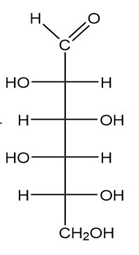
What do you think about this solution?
We value your feedback to improve our textbook solutions.