Chapter 29: Q2P (page 1156)
What form exists at the isoelectric point of each of the following amino acids: (a) valine; (b)leucine; (c)proline; (d) glutamic acid
Short Answer
Answer
a.
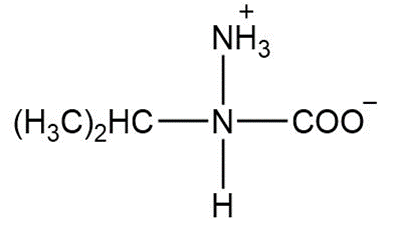
b.
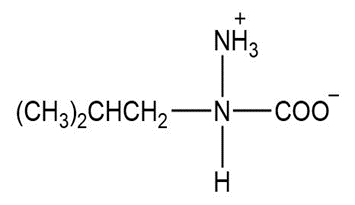
c.
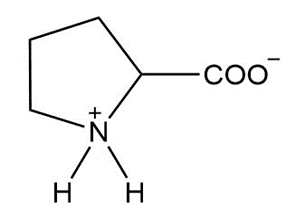
d.
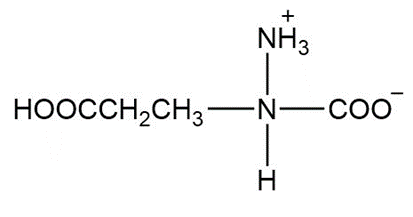
Learning Materials
Features
Discover
Chapter 29: Q2P (page 1156)
What form exists at the isoelectric point of each of the following amino acids: (a) valine; (b)leucine; (c)proline; (d) glutamic acid
Answer
a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why the pKaof the -NH3+group of an -amino acid is lower than the pKaof the ammonium ion derived from a amine . For example, pKaof the group of alanine is 9.87 but the pKaof is 10.63.
Devise a synthesis of the following dipeptide from amino acid starting materials.
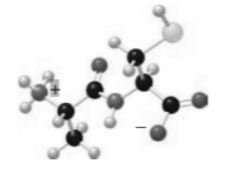
Tryptophan is not classified as a basic amino acid even though it has a heterocycle containing a nitrogen atom. Why is the N atom in the five-membered ring of tryptophan not readily protonated by acid?
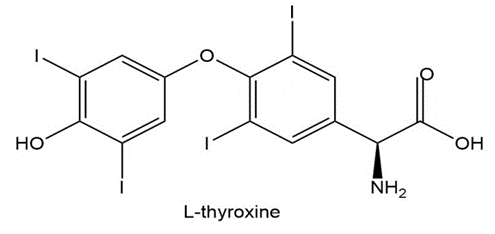
L-thyroxine, a thyroid hormone and oral medication used to treat thyroid hormone deficiency, is an amino acid that does not exist in proteins. Draw the zwitterionic form of L-thyroxine.
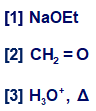
What amino acid is formed when CH3CONHCH(CO2Et)2is treated with the following series of reagents:
What do you think about this solution?
We value your feedback to improve our textbook solutions.