Chapter 29: Q1P (page 1155)
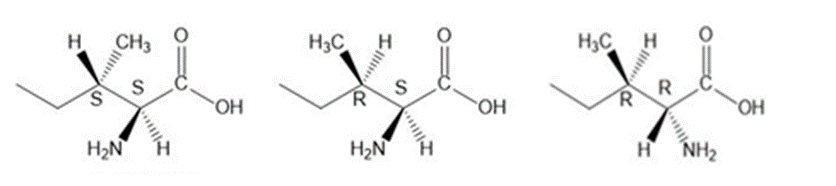
Draw the other three stereoisomers of L-isoleucine, and label the stereogenic centers as R or S.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 29: Q1P (page 1155)
Draw the other three stereoisomers of L-isoleucine, and label the stereogenic centers as R or S.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
What types of stabilizing interactions exist between each of the following pairs of amino acids?
a. Ser and Tyr
b. Val and Leu
c. two Phe residues
Histidine is classified as a basic amino acid because one of the N atoms in its five-membered ring is readily protonated by acid. Which N atom in histidine is protonated and why?
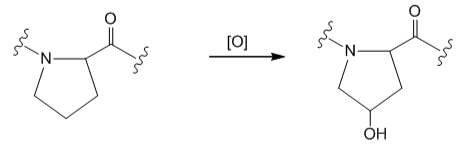
After the peptide chain of collage has been formed, many of the proline residues are hydroxylated on one of the ring carbon atoms. Why is this process important for the triple helix of collagen?
What alkyl halide is needed to synthesize each amino acid from diethyl acetamidomalonate: (a)Asn; (b)His; (c)Trp?
(a) What products are formed when each peptide is treated with trypsin? (b) What products are formed when each peptide is treated with chymotrypsin?
[1] Gly–Ala–Phe–Leu–Lys–Ala
[2] Phe–Tyr–Gly–Cys–Arg–Ser
[3] Thr–Pro–Lys–Glu–His–Gly–Phe–Cys–Trp–Val–Val–Phe
What do you think about this solution?
We value your feedback to improve our textbook solutions.