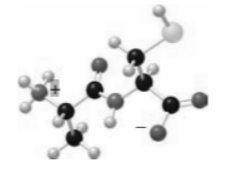
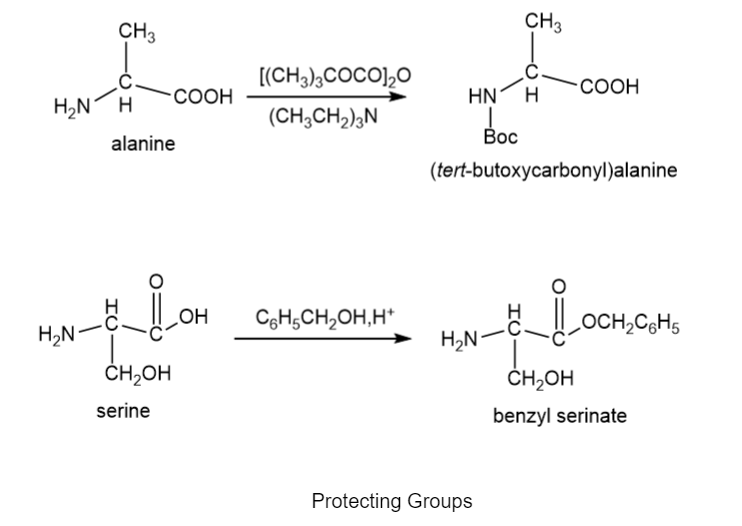
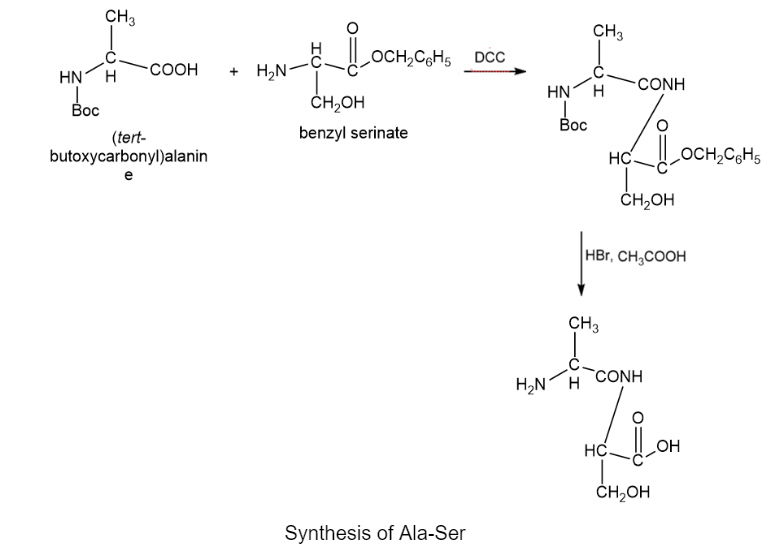
Chapter 29: 24P (page 1177)
Devise a synthesis of the following dipeptide from amino acid starting materials.
Short Answer
Learning Materials
Features
Discover
Chapter 29: 24P (page 1177)
Devise a synthesis of the following dipeptide from amino acid starting materials.



All the tools & learning materials you need for study success - in one app.
Get started for free
What is the structure of each amino acid at its isoelectric point: (a) alanine (b) methionine; (c)aspartic acid; (d) lysine?
Draw the organic products formed in each reaction

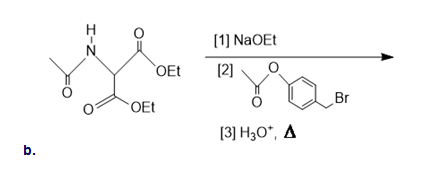
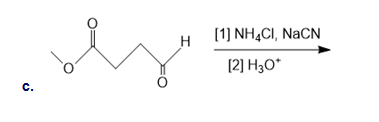
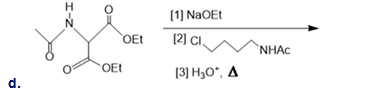
Draw the organic products formed in the following reaction.
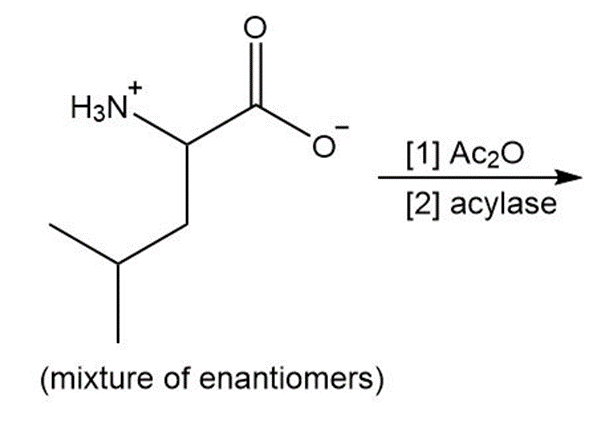
After the peptide chain of collage has been formed, many of the proline residues are hydroxylated on one of the ring carbon atoms. Why is this process important for the triple helix of collagen?
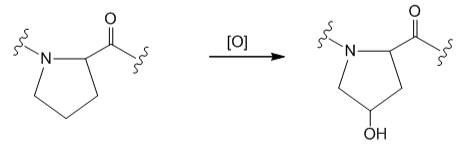
Draw the structure of each peptide. Label the N-terminal and C-terminal amino acids and all amide bonds.
a. Val–Glu
b. Gly–His–Leu
c. M–A–T–T
What do you think about this solution?
We value your feedback to improve our textbook solutions.