Chapter 29: Q35P (page 1192)
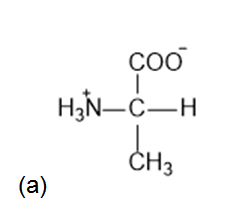
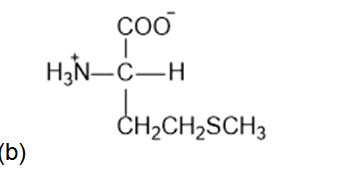
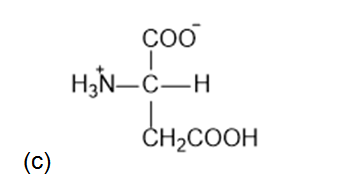
What is the structure of each amino acid at its isoelectric point: (a) alanine (b) methionine; (c)aspartic acid; (d) lysine?
Short Answer

Learning Materials
Features
Discover
Chapter 29: Q35P (page 1192)
What is the structure of each amino acid at its isoelectric point: (a) alanine (b) methionine; (c)aspartic acid; (d) lysine?




All the tools & learning materials you need for study success - in one app.
Get started for free
Consider two molecules of a tetrapeptide composed of only alanine residues. Draw the hydrogen bonding interactions that result when these two peptides adopt a parallel β-pleated sheet arrangement. Answer this same question for the antiparallel β-pleated sheet arrangement.
Explain why the pKaof the -NH3+group of an -amino acid is lower than the pKaof the ammonium ion derived from a amine . For example, pKaof the group of alanine is 9.87 but the pKaof is 10.63.
Deduce the sequence of a heptapeptide that contains the amino acids Ala, Arg, Glu, Gly, Leu, Phe, and Ser, from the following experimental data. Edman degradation cleaves Leu from the heptapeptide, and carboxypeptidase forms Glu and a hexapeptide. Treatment of the heptapeptide with chymotrypsin forms a hexapeptide and a single amino acid. Treatment of the heptapeptide with trypsin forms a pentapeptide and a dipeptide. Partial hydrolysis forms Glu, Leu, Phe, and the tripeptidesGly–Ala–Ser and Ala–Ser–Arg
Draw the organic products formed in each reaction.
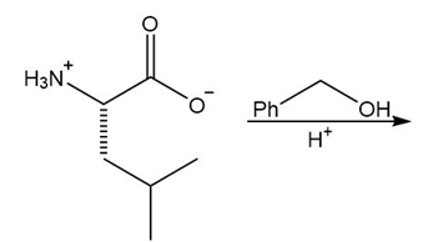
a.
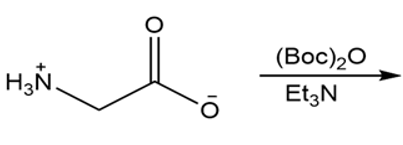
b.

c.
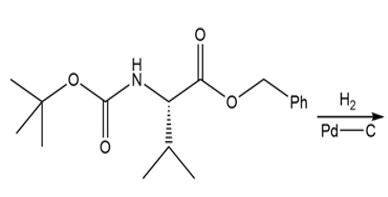
d.
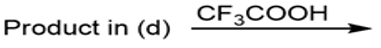
e.

f.
What is the predominant form of each of the following amino acids at pH=11? What is the overall charge on the amino acid? (a)valine; (b)proline; (c)glutamic acid; (d)lysine?
What do you think about this solution?
We value your feedback to improve our textbook solutions.