Chapter 29: Q60P (page 1196)
Draw the organic products formed in each reaction.
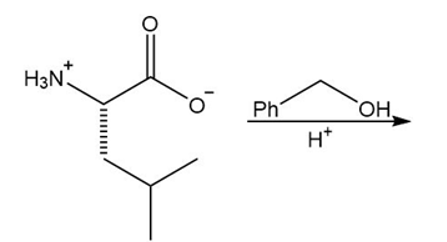
a.
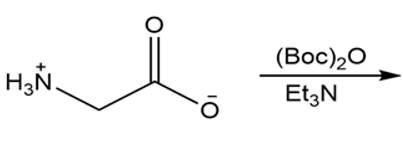
b.

c.
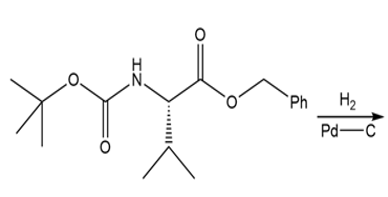
d.
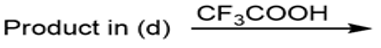
e.
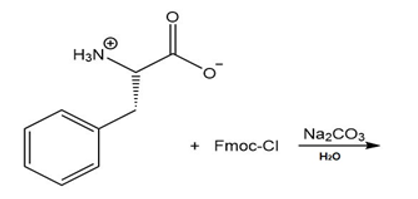
f.
Short Answer
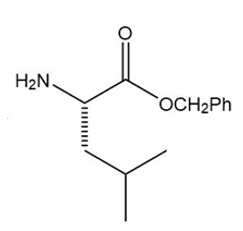
a.
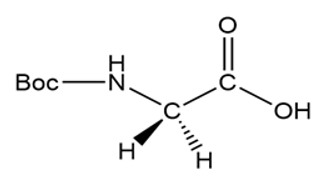
b.
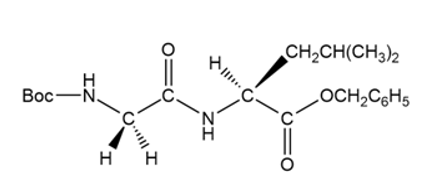
c.
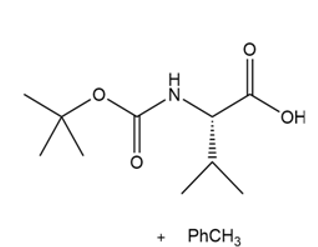
d.
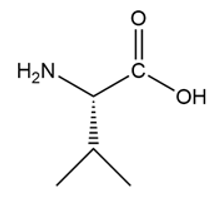
e.
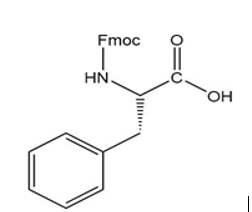
f.
Learning Materials
Features
Discover
Chapter 29: Q60P (page 1196)
Draw the organic products formed in each reaction.

a.

b.

c.

d.
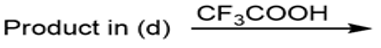
e.

f.

a.

b.

c.

d.

e.

f.
All the tools & learning materials you need for study success - in one app.
Get started for free
Histidine is classified as a basic amino acid because one of the N atoms in its five-membered ring is readily protonated by acid. Which N atom in histidine is protonated and why?
Glutathione, a powerful antioxidant that destroys harmful oxidizing agents in cells, is composed of glutamic acid, cysteine, and glycine, and has the following structure:
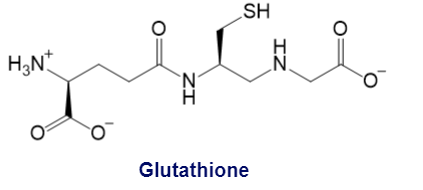
a. What product is formed when glutathione reacts with an oxidizing agent?
b. What is unusual about the peptide bond between glutamic acid and cysteine?
An octapeptide contains the following amino acids: Arg, Glu, His, Ile, Leu, Phe, Tyr, and Val. Carboxypeptidase treatment of the octapeptide forms Phe and a heptapeptide. Treatment of the octapeptide with chymotrypsin forms two tetrapeptides, A and B. Treatment of A with trypsin yields two dipeptides, C and D. Edman degradation cleaves the following amino acids from each peptide: Glu (octapeptide), Glu (A), Ile (B), Glue (C), and Val (D). Partial hydrolysis of tetrapeptide B forms Ile-Leu in addition to other products. Deduce the structure of the octapeptide and fragments A-D.
Tryptophan is not classified as a basic amino acid even though it has a heterocycle containing a nitrogen atom. Why is the N atom in the five-membered ring of tryptophan not readily protonated by acid?
What is the predominant form of each of the following amino acids at pH=1? What is the overall charge on the amino acid at this pH?(a)threonine ; (b)methionine; (c)aspartic acid; (d)arginine
What do you think about this solution?
We value your feedback to improve our textbook solutions.