Chapter 29: Q31. (page 1192)
Devise a synthesis of the following dipeptide from amino acid starting materials.
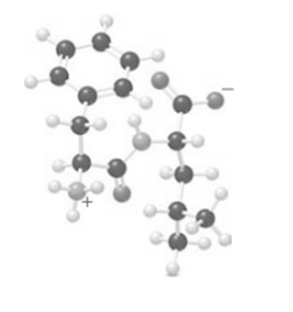
Short Answer
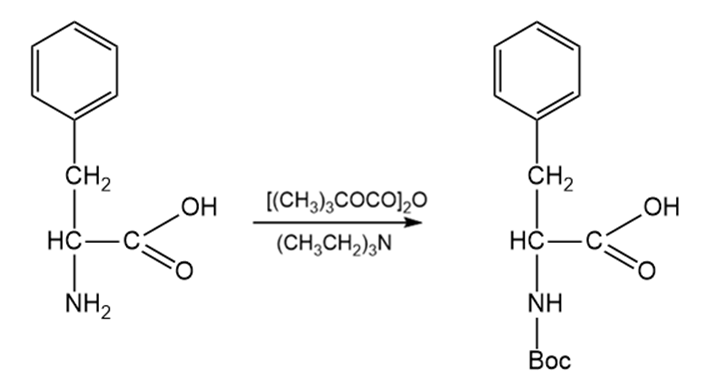
Protecting Amine Group
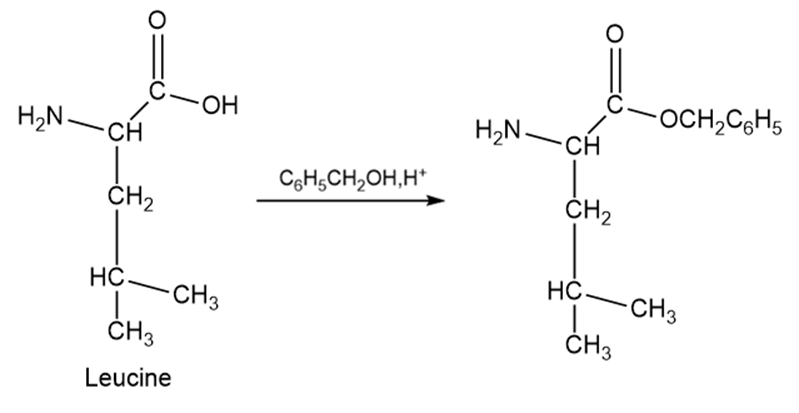
Protecting –COOH group.
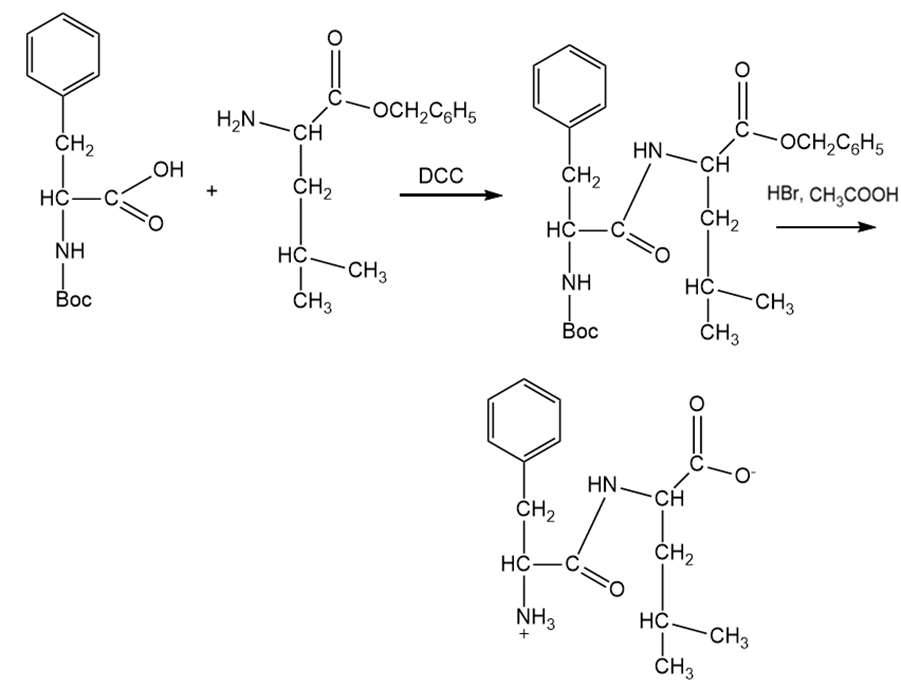
Synthesis of Phe-Leu.
Learning Materials
Features
Discover
Chapter 29: Q31. (page 1192)
Devise a synthesis of the following dipeptide from amino acid starting materials.


Protecting Amine Group

Protecting –COOH group.

Synthesis of Phe-Leu.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of each peptide. Label the N-terminal and C-terminal amino acids and all amide bonds.
a. Val–Glu
b. Gly–His–Leu
c. M–A–T–T
What aldehyde is needed to synthesize each amino acid by the Strecker synthesis?
(a) Valine;
(b) Leucine;
(c) Phenylalanine.
What types of stabilizing interactions exist between each of the following pairs of amino acids?
a. Ser and Tyr
b. Val and Leu
c. two Phe residues
What is the predominant form of each of the following amino acids at pH=11? What is the overall charge on the amino acid? (a)valine; (b)proline; (c)glutamic acid; (d)lysine?
What is the predominant form of each of the following amino acids at pH=1? What is the overall charge on the amino acid at this pH?(a)threonine ; (b)methionine; (c)aspartic acid; (d)arginine
What do you think about this solution?
We value your feedback to improve our textbook solutions.