Chapter 21: 79P (page 817)
Draw the structure of the acyclic polyhydroxy aldehyde that cyclizes to each hemiacetal.
a.
Short Answer
a
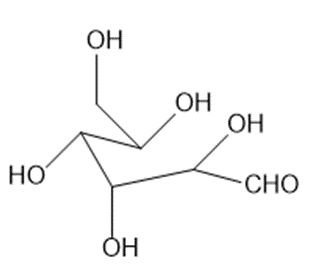
Acyclic polyhydroxy aldehyde of a.
b
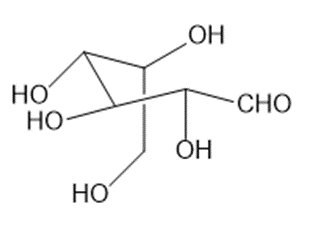
Acyclic polyhydroxy aldehyde of b.
Learning Materials
Features
Discover
Chapter 21: 79P (page 817)
Draw the structure of the acyclic polyhydroxy aldehyde that cyclizes to each hemiacetal.
a.
a

Acyclic polyhydroxy aldehyde of a.
b

Acyclic polyhydroxy aldehyde of b.
All the tools & learning materials you need for study success - in one app.
Get started for free
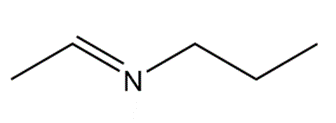
What 1° amine and carbonyl compound are needed to prepare each imine?
a.
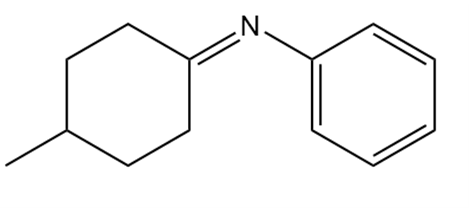
b.
Draw the products formed in each reaction sequence.
a.
b.
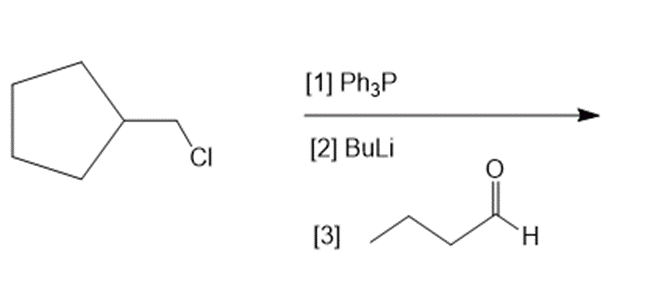
What reagents are needed to convert each compound into butanal?
a.
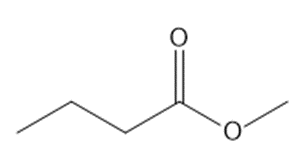
b.

c.

d.
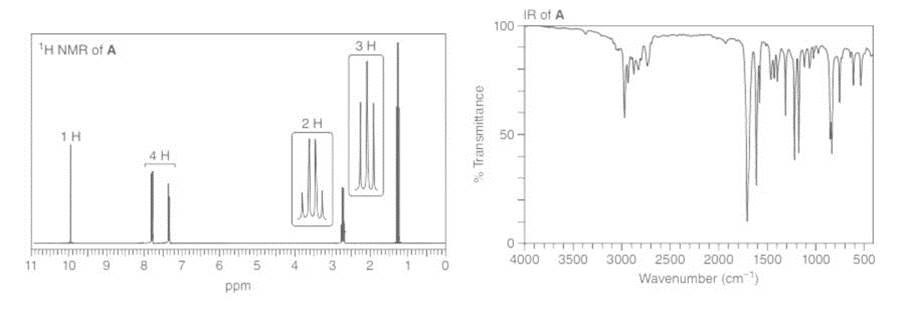
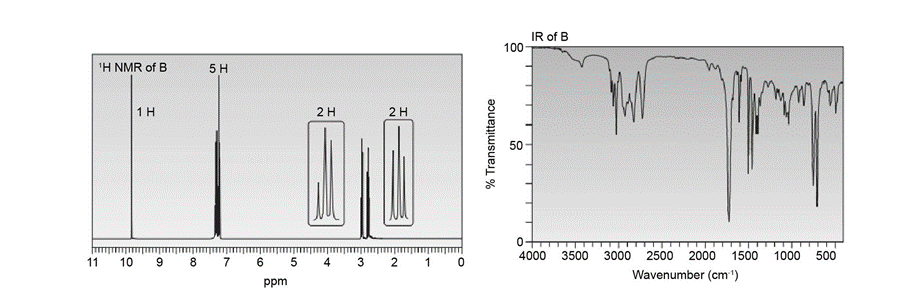
Compounds A and B have molecular formula C9H10O . Identify their structures from the 1HNMR and IR spectra given.
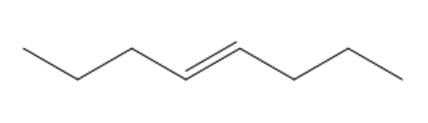
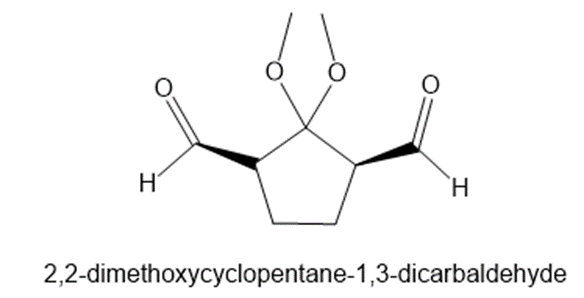
What alkene would yield 2,2-dimethoxycyclopentane-1,3-dicarbaldehyde on treatment withfollowed by ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.