Chapter 21: 45P (page 817)
Draw the products formed in each reaction sequence.
a.
b.
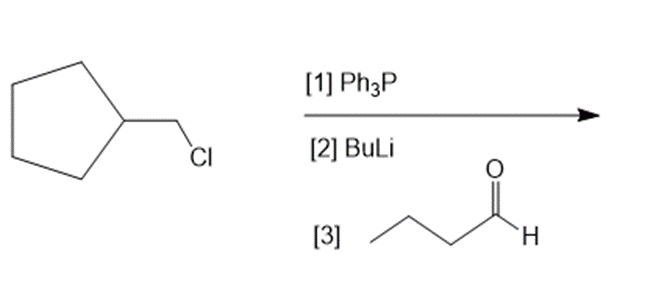
Short Answer
a.
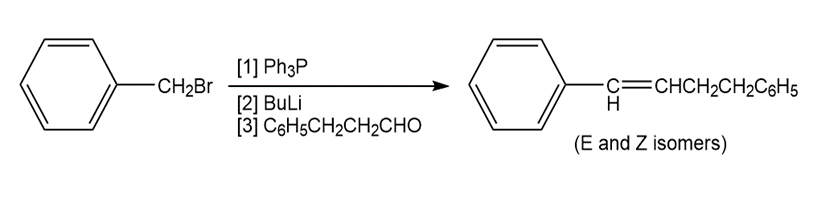
b.
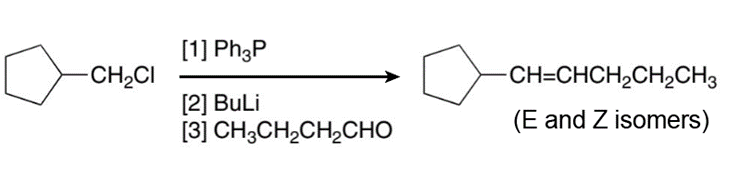
Learning Materials
Features
Discover
Chapter 21: 45P (page 817)
Draw the products formed in each reaction sequence.
a.
b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
What Wittig reagent and carbonyl compound are needed to prepare each alkene? When two routes are possible, indicate which route, if any, is preferred.
a.
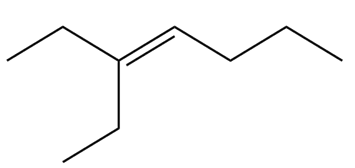
b.
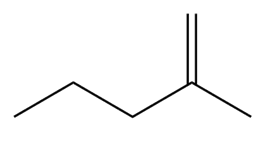
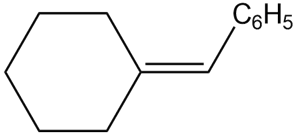
c.
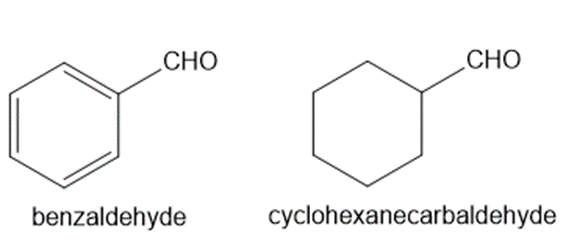
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
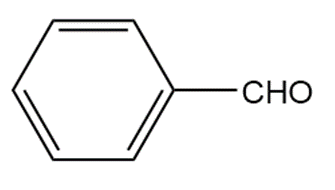
What cyanohydrin and carbonyl compound are formed when amygdalin is metabolized in a similar manner to linamarin?
�ٰ����� t��� p���ǻ�ܳ��� f�ǰ������ w���� C��3CH2CH2CH2NH2reacts with each carbonyl compound in the presence of mild acid.
a.
b.
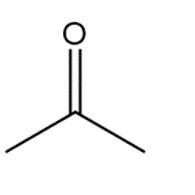
c.
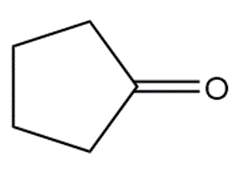
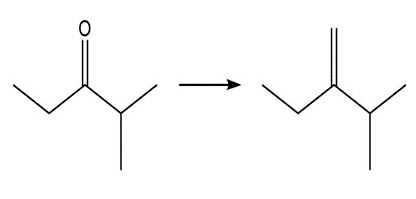
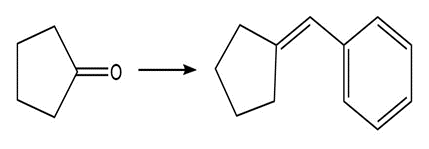
Show two methods to synthesize each alkene: a one-step method using a Wittig reagent, and a two-step method that forms a carbon–carbon bond with an organometallic reagent in one of the steps.
a.
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.