Chapter 20: Q6E (page 1139)
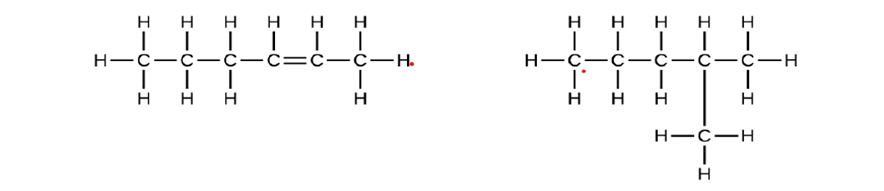
Explain why these two molecules are not isomers:
Short Answer
They are not isomers since their molecular formulas differ.
Learning Materials
Features
Discover
Chapter 20: Q6E (page 1139)
Explain why these two molecules are not isomers:

They are not isomers since their molecular formulas differ.
All the tools & learning materials you need for study success - in one app.
Get started for free
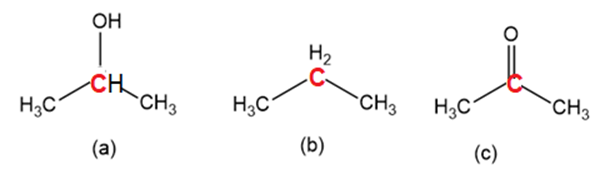
Order the following molecules from least to most oxidized, based on the marked carbon atom:
Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
Ethylammonium chloride is added to a solution of sodium hydroxide.
Write Lewis structures and describe the molecular geometry of each carbon atom in the following compounds:
(a) cis-3-hexene
(b) cis-1-chloro-2-bromoethene
(c) 2-pentyne
(d)trans-6-ethyl-7-methyl-2-octene
Benzene is one of the compounds used as an octane enhancer in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2H2 C6H6
Draw Lewis structures for these compounds, with resonance structures as appropriate, and determine the hybridization of the carbon atoms in each.
Butane is used as a fuel in disposable lighters. Write the Lewis structure for each isomer of butane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.