Chapter 20: Q60E (page 1146)
Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
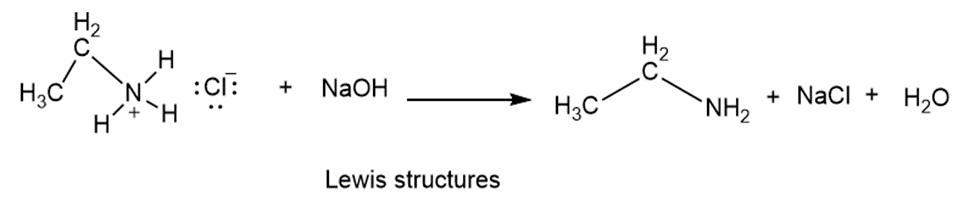
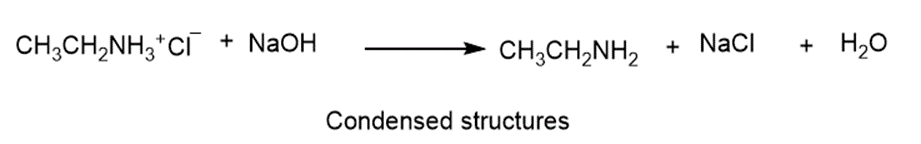
Ethylammonium chloride is added to a solution of sodium hydroxide.
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q60E (page 1146)
Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
Ethylammonium chloride is added to a solution of sodium hydroxide.


All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why it is not possible to prepare a ketone that contains only two carbon atoms.
Write two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
(a) propanol is converted to dipropyl ether
(b) propene is treated with water in dilute acid.
Give the complete IUPAC name for each of the following compounds:
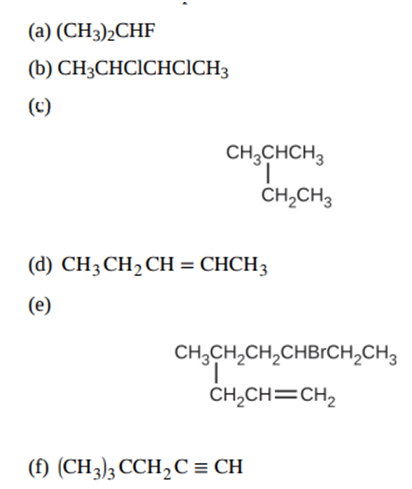
Alcohols\(A,B\) , and \(C\) all have the composition \({C_4}{H_{10}}O.\) Molecules of alcohol A contain a branched carbon chain and can be oxidized to an aldehyde; molecules of alcohol B contain a linear carbon chain and can be oxidized to a ketone; and molecules of alcohol C can be oxidized to neither an aldehyde nor a ketone. Write the Lewis structures of these molecules.
Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
(a) 2-butene reacts with chlorine.
(b) benzene burns in air.
What do you think about this solution?
We value your feedback to improve our textbook solutions.