Chapter 20: Q7E (page 1139)
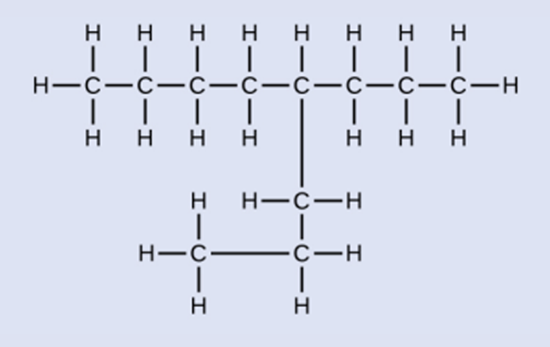
Explain why these two molecules are not isomers:
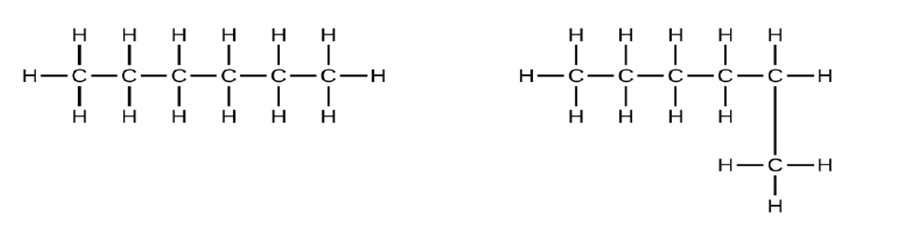
Short Answer
Because they are the same compound, they are not isomers.
Learning Materials
Features
Discover
Chapter 20: Q7E (page 1139)
Explain why these two molecules are not isomers:

Because they are the same compound, they are not isomers.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw Lewis structures for pyridine and its conjugate acid, the pyridinium ion, \({C_5}{H_5}N{H^ + }.\) \(What are\) the geometries and hybridizations about the nitrogen atoms in pyridine and in the pyridinium ion?
Give the complete IUPAC name and the common name for each of the following compounds:
a)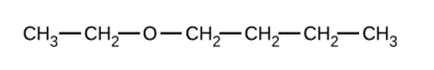
b)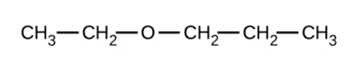
c)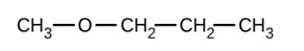
Ethylene can be produced by the pyrolysis of ethane:
\[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{6}}}\to{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{4}}}{\rm{+}}{{\rm{H}}_{\rm{2}}}\]
How many kilograms of ethylene is produced by the pyrolysis of 1.000 ×103 kg of ethane, assuming a 100.0%yield?
Write the structures for all the isomers of the \(--{C_5}{H_{11}}\) alkyl group.
Name the following molecule:
What do you think about this solution?
We value your feedback to improve our textbook solutions.