Chapter 20: Q40 E (page 1143)
Order the following molecules from least to most oxidized, based on the marked carbon atom:
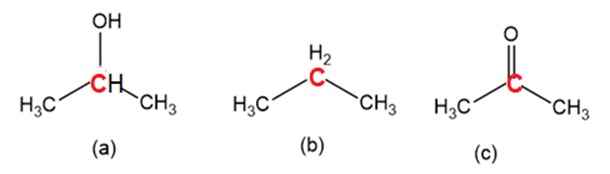
Short Answer
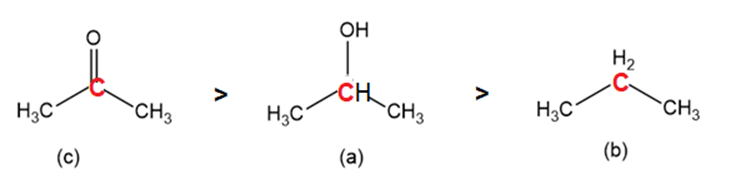
Learning Materials
Features
Discover
Chapter 20: Q40 E (page 1143)
Order the following molecules from least to most oxidized, based on the marked carbon atom:


All the tools & learning materials you need for study success - in one app.
Get started for free
Name the following molecule:
Isooctane is the common name of the isomer of\({C_8}{H_{18}}\)used as the standard of 100 for the gasoline octane rating:
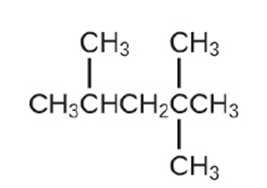
(a) What is the IUPAC name for the compound?
(b) Name the other isomers that contain a five-carbon chain with three methyl substituents.
Write two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
(a) 1-butanol reacts with acetic acid
(b) propionic acid is poured onto solid calcium carbonate
Give the complete IUPAC name for each of the following compounds:
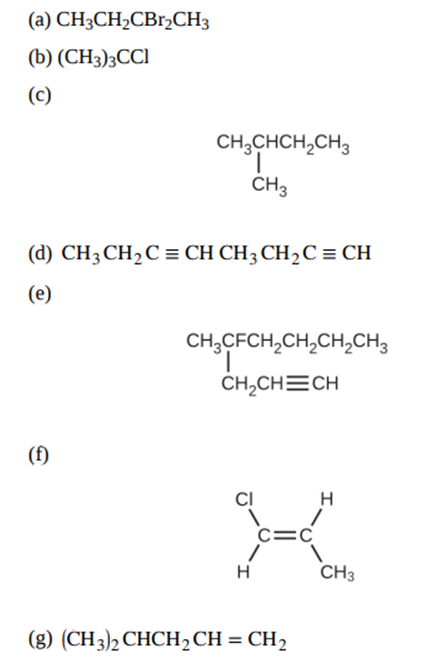
Write the Lewis structures of all isomers with the formula \({C_3}{H_7}ON \) that contain an amide linkage.
What do you think about this solution?
We value your feedback to improve our textbook solutions.