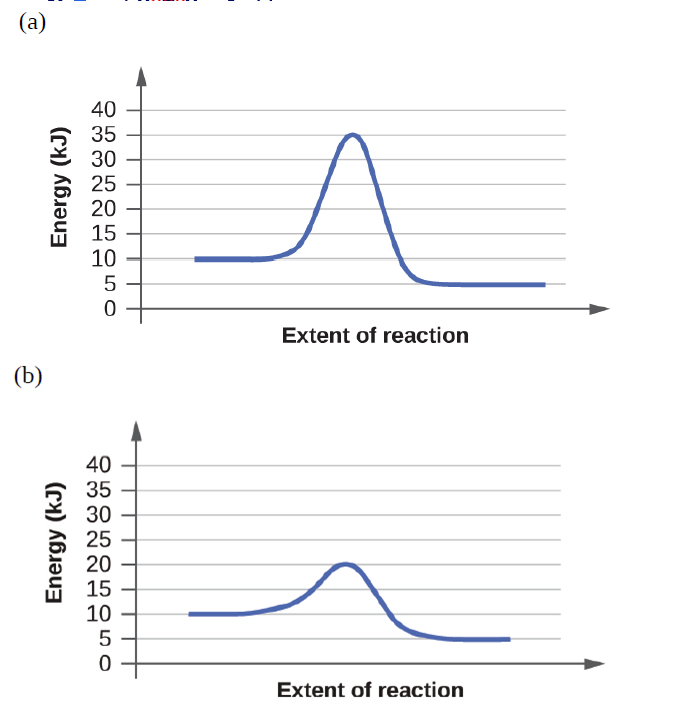
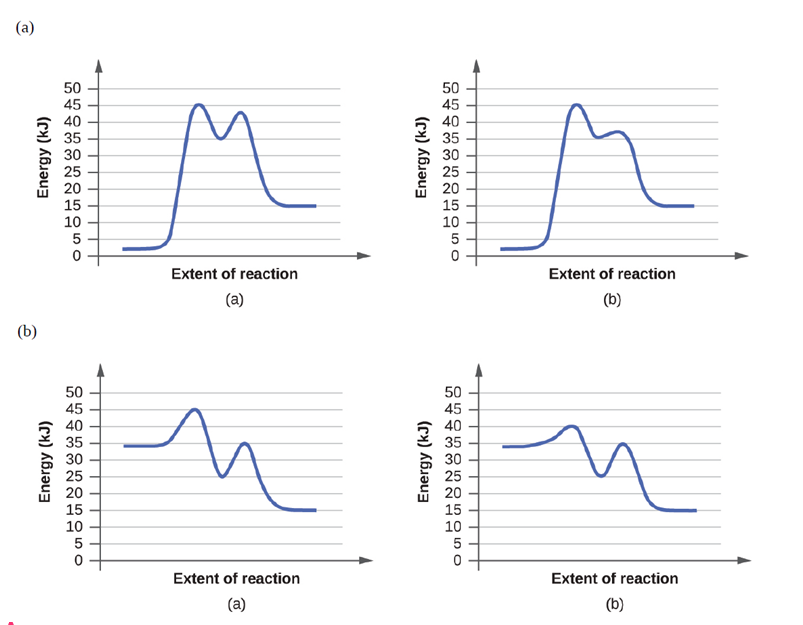
Chapter 12: Q9E (page 702)
Go to the PhET Reactions & Rates interactive. Use the Single Collision tab to represent how the collision between monatomic oxygen (O) and carbon monoxide (CO) results in the breaking of one bond and the formation of another. Pull back on the red plunger to release the atom and observe the results. Then, click on “Reload Launcher” and change to “Angled shot” to see the difference.
- What happens when the angle of the collision is changed?
- Explain how this is relevant to rate of reaction.
Short Answer
- When angle of collision is changed it affects the formation of bonds and breaking of bonds between monoatomic oxygen and carbon monoxide.
- Angle of collision increases or decreases the rate of reaction.