At different temperatures, say \({{\bf{T}}_{\bf{1}}}\), \({{\bf{T}}_{\bf{2}}}\), and \({{\bf{T}}_3}\); let the rate of the reaction is denoted by \({{\bf{R}}_{\bf{1}}}\), \({{\bf{R}}_{\bf{2}}}\) and \({{\bf{R}}_{\bf{3}}}\).
Thus, the equation becomes,
\(\begin{align}{}\log \,{R_1} = \log \,Z - 2.303\frac{{{E_a}}}{{R{T_1}}}\\\log \,{R_2} = \log \,Z - 2.303\frac{{{E_a}}}{{R{T_2}}}\\\log \,{R_3} = \log \,Z - 2.303\frac{{{E_a}}}{{R{T_3}}}\end{align}\)
Linear plot for reaction rates at different temperature is shown as:
![]()
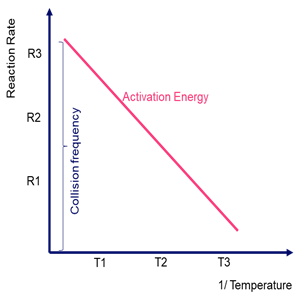
Comparing this to the linear equation,
\({\bf{y = mx + c}}\)
where, m and c represent the slope and intercept in the graph. Herein, the slope gives the activation energy value
Hence, the slope of this linear plot will provide the value for the activation energy of the reaction.




