Chapter 9: Gases
Q15E
The pressure of a sample of gas is measured at sea level with an open-end mercury manometer. Assuming atmospheric pressure is 760.0mm Hg, determine the pressure of the gas in (a) mm Hg (b) (c) kPa.
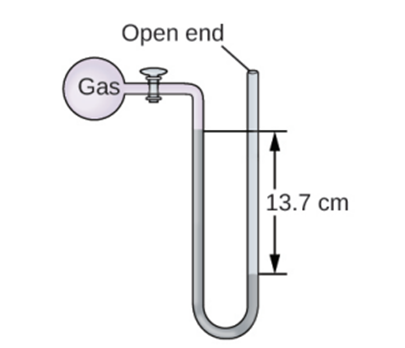
Q16 E
The pressure of a sample of gas is measured at sea level with an open-end mercury manometer. Assuming atmospheric pressure is 760.0mm Hg ,determine the pressure of the gas in: (a) mm Hg (b) (c) kPa.
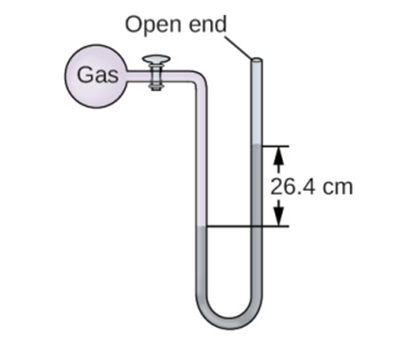
Q17 E
How would the use of a volatile liquid affect the measurement of gas using open-ended manometers vs. closed-end manometers?
Q18 E
Sometimes leaving a bicycle in the sun on a hot day will cause a blowout. Why?
Q19 E
Explain how the volume of the bubbles exhausted by a scuba diver ( Figure 9.6 ) change as they rise to the surface, assuming that they remain intact.
Q1 E
Why are sharp knives more effective than dull knives (Hint: think about the definition of pressure)?
Q20 E
One way to state Boyle’s law is “All other things being equal, the pressure of a gas is inversely proportional to its volume.” (a) What is the meaning of the term “inversely proportional?” (b) What are the “other things” that must be equal?
Q21 E
An alternate way to state Avogadro’s law is “All other things being equal, the number of molecules in a gas is directly proportional to the volume of the gas.” (a) What is the meaning of the term “directly proportional?” (b) What are the “other things” that must be equal?
Q22 E
How would the graph in Figure 9.12 change if the number of moles of gas in the sample used to determine the curve were doubled?
Q23E
How would the graph in Figure \({\rm{9}}{\rm{.13}}\) change if the number of moles of gas in the sample used to determine the curve were doubled?
