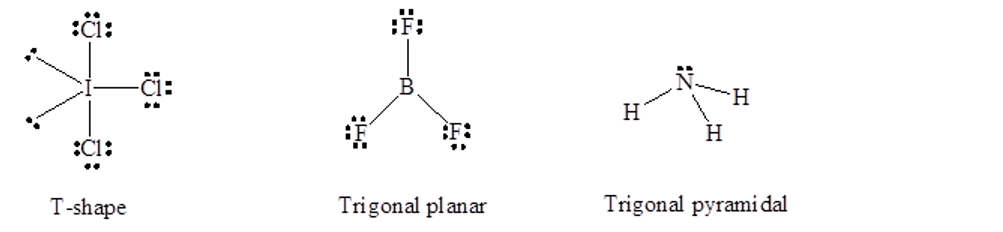
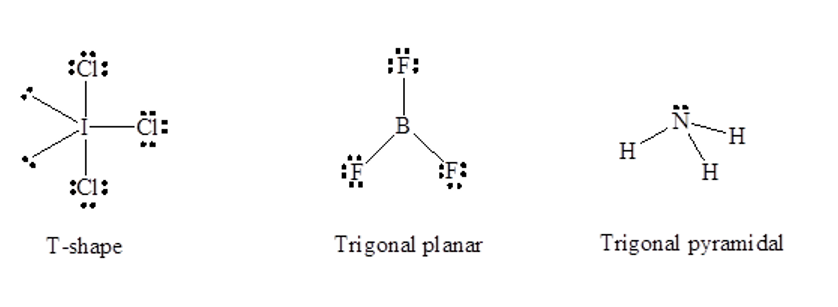
Chapter 7: Q108E (page 405)
A molecule with the formula\({\rm{A}}{{\rm{B}}_{\rm{3}}}\), in which A and B represent different atoms, could have one of three different shapes. Sketch and name the three different shapes that this molecule might have. Give an example of a molecule or ion that has each shape.
Short Answer
A molecule with molecular formula \({\rm{A}}{{\rm{B}}_{\rm{3}}}\) can have T-shape for example \({\rm{IC}}{{\rm{l}}_{\rm{3}}}\), trigonal planar for example \({\rm{B}}{{\rm{F}}_{\rm{3}}}\)and trigonal pyramidal for example \({\rm{N}}{{\rm{H}}_{\rm{3}}}\) shapes shown as follows: