Chapter 7: Q1E (page 389)
Does a cation gain protons to form a positive charge or does it lose electrons?
Short Answer
To form cations and anions, atoms gain and lose electrons, while their proton count remains constant.
Learning Materials
Features
Discover
Chapter 7: Q1E (page 389)
Does a cation gain protons to form a positive charge or does it lose electrons?
To form cations and anions, atoms gain and lose electrons, while their proton count remains constant.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use principles of atomic structure to answer each of the following:
(a) The radius of the \({\rm{Ca}}\) atom is \({\rm{197 pm}}\); the radius of the \({\rm{C}}{{\rm{a}}^{{\rm{2 + }}}}\) ion is \({\rm{99 pm}}\). Account for the difference.
(b) The lattice energy of \({\rm{CaO(s)}}\) is \({\rm{ - 3460 kJ/mol}}\); the lattice energy of \({{\rm{K}}_{\rm{2}}}{\rm{O}}\) is \({\rm{ - 2240 kJ/mol}}\). Account for the difference.
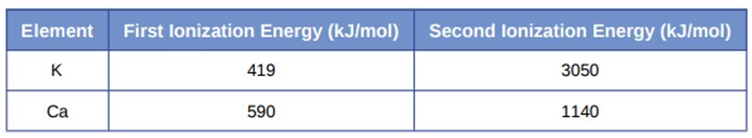
(c) Given these ionization values, explain the difference between \({\rm{Ca}}\) and \({\rm{K}}\) with regard to their first and second ionization energies.
(d) The first ionization energy of \({\rm{Mg}}\) is \({\rm{738 kJ/mol}}\) and that of \({\rm{Al}}\) is \({\rm{578 kJ/mol}}\). Account for this difference.
Question: Calculate the formal charge of each element in the following compounds and ions:
(a) \({{\rm{F}}_{\rm{2}}}{\rm{CO}}\)
(b) \({\rm{N}}{{\rm{O}}^{\rm{ - }}}\)
(c) \({\rm{BF}}_{\rm{4}}^{\rm{ - }}\)
(d) \({\rm{SnCl}}_{\rm{3}}^{\rm{ - }}\)
(e) \({{\rm{H}}_{\rm{2}}}{\rm{CC}}{{\rm{H}}_{\rm{2}}}\)
(f) \({\rm{Cl}}{{\rm{F}}_{\rm{3}}}\)
(g) \({\rm{Se}}{{\rm{F}}_{\rm{6}}}\)
(h) \({\rm{PO}}_{\rm{4}}^{{\rm{3 - }}}\)
Which compound in each of the following pairs has the larger lattice energy? Note: \({\rm{B}}{{\rm{a}}^{{\rm{2 + }}}}\) and \({{\rm{K}}^{\rm{ + }}}\) have similar radii; \({{\rm{S}}^{{\rm{2 - }}}}\) and \({\rm{C}}{{\rm{l}}^{\rm{ - }}}\) have similar radii. Explain your choices.
(a) \({{\rm{K}}_{\rm{2}}}{\rm{O}}\) or \({\rm{N}}{{\rm{a}}_{\rm{2}}}{\rm{O}}\)
(b) \({{\rm{K}}_{\rm{2}}}{\rm{S}}\) or \({\rm{BaS}}\)
(c) \({\rm{KCl}}\) or \({\rm{BaS}}\)
(d) \({\rm{BaS}}\) or \({\rm{BaC}}{{\rm{l}}_{\rm{2}}}\)
Which of the following compounds requires the most energy to convert one mole of the solid into separate ions?
(a) \({\rm{MgO}}\)
(b) \({\rm{SrO}}\)
(c) \({\rm{KF}}\)
(d) \({\rm{CsF}}\)
(e) \({\rm{Mg}}{{\rm{F}}_{\rm{2}}}\)
From its position in the periodic table, determine which atom in each pair is more electronegative: (a)\({\rm{N or P}}\)(b)\({\rm{N or Ge}}\)(c)\({\rm{S or F}}\)(d)\({\rm{Cl or S}}\)(e)\({\rm{H or C}}\)(f)\({\rm{Se or P}}\)(g)\({\rm{C or Si}}\).
What do you think about this solution?
We value your feedback to improve our textbook solutions.