Chapter 7: Q20E (page 392)
Which is the most polar bond? (a) \({\rm{C - C}}\) (b) \({\rm{C - H}}\) (c) \({\rm{N - H}}\) (d) \({\rm{O - H}}\) (e) \({\rm{Se - H}}\) .
Short Answer
The most polar bond is: \({\rm{O - H}}\).
Learning Materials
Features
Discover
Chapter 7: Q20E (page 392)
Which is the most polar bond? (a) \({\rm{C - C}}\) (b) \({\rm{C - H}}\) (c) \({\rm{N - H}}\) (d) \({\rm{O - H}}\) (e) \({\rm{Se - H}}\) .
The most polar bond is: \({\rm{O - H}}\).
All the tools & learning materials you need for study success - in one app.
Get started for free
A molecule with the formula\({\rm{A}}{{\rm{B}}_{\rm{2}}}\), in which A and B represent different atoms, could have one of three different shapes. Sketch and name the three different shapes that this molecule might have. Give an example of a molecule or ion for each shape.
The arrangement of atoms in several biologically important molecules is given here. Complete the Lewis structures of these molecules by adding multiple bonds and lone pairs. Do not add any more atoms.
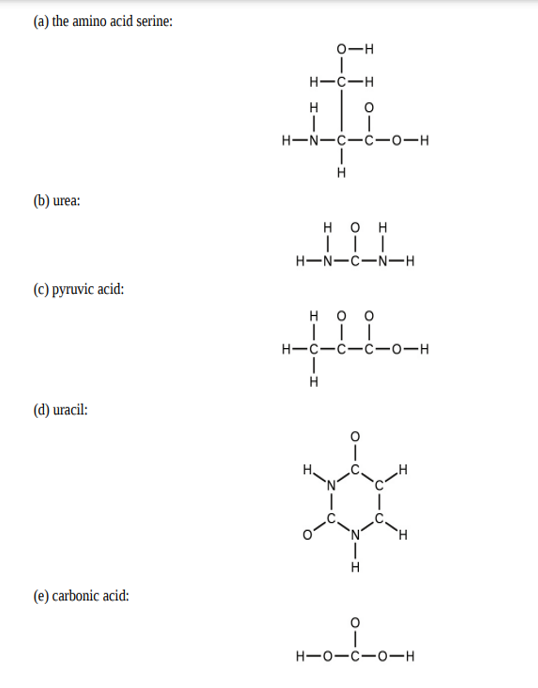
What feature of a Lewis structure can be used to tell if a molecule’s (or ion’s) electron-pair geometry and molecular structure will be identical?
What is the molecular structure of the stable form of \({\rm{FN}}{{\rm{O}}_{\rm{2}}}\) ? (\({\rm{N}}\)is the central atom.)
Which of the following molecules and ions contain polar bonds? Which of these molecules and ions have dipole moments?
What do you think about this solution?
We value your feedback to improve our textbook solutions.