Chapter 2: Atoms, Molecules, and Ions
Q27 E
Explain why the symbol for an atom of the element oxygen and the formula for a molecule of oxygen differ.
Q28 E
Explain why the symbol for element sulfur and the formula for a molecule of sulfur differ.
Q29E
Write the molecular and empirical formulas of the following compounds:\(\begin{aligned}{}a)O = C = O\\b)H - C \equiv C - H\end{aligned}\)
c)
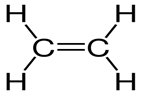
(d)
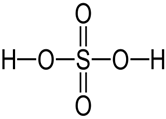
Q2 E
Which postulate of Dalton’s theory is consistent with the following observation concerning the weights of reactants and products? When 100 grams of solid calcium carbonate is heated, 44 grams of carbon dioxide and 56 grams of calcium oxide are produced.
Q30E
Write the molecular and empirical formulas of the following compounds:
a)
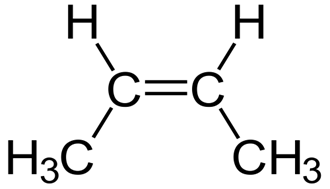
(b)
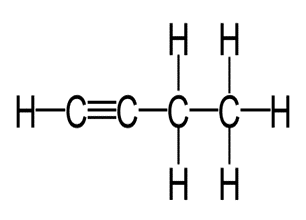
(c)
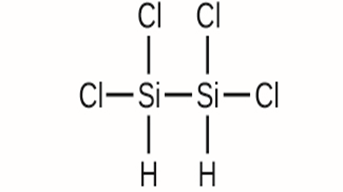
(d)
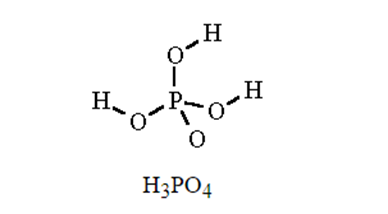
Q.31E
Determine the empirical formula for the following compounds:
a) caffeine, C6H10N4O2
b) fructose, C12H22O11
c) hydrogen peroxide, H2O2
d) glucose, C6H12O6
e) ascorbic acid (vitamin C), C6H8O6
Q.32E
Determine the empirical formulas for the following compounds.
a. acetic acid, C2H4O2
b. citric acid, C6H8O7
c. hydrazine, N2H4
d. nicotine, C10H14N2
e. butane, C4H10
Q.33E
Write the empirical formula for the following compounds:
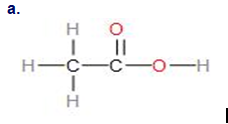
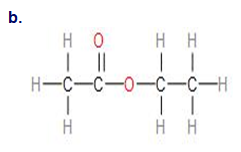
Q.34E
Open the Build a Molecule simulation (http://openstaxcollege.org/l/16molbuilding) and select the “Larger Molecules” tab. Select an appropriate atoms “Kit” to build a molecule with two carbon and six hydrogen atoms. Drag atoms into the space above the “Kit” to make a molecule. A name will appear when you have made an actual molecule that exists (even if it is not the one you want). You can use the scissors tool to separate atoms if you would like to change the connections. Click on “3D” to see the molecule, and look at both the space-filling and ball and-stick possibilities.(a) Draw the structural formula of this molecule and state its name.(b) Can you arrange these atoms in any way to make a different compound?
Q.35E
Use the Build a Molecule simulation (http://openstaxcollege.org/l/16molbuilding) to repeat Exercise 2.34, but build a molecule with two carbons, six hydrogens, and one oxygen.(a) Draw the structural formula of this molecule and state its name.(b) Can you arrange these atoms to make a different molecule? If so, draw its structural formula and state its name.(c) How are the molecules drawn in (a) and (b) the same? How do they differ? What are they called (the type of relationship between these molecules, not their names)?
