Chapter 2: Q28 E (page 128)
Explain why the symbol for element sulfur and the formula for a molecule of sulfur differ.
Learning Materials
Features
Discover
Chapter 2: Q28 E (page 128)
Explain why the symbol for element sulfur and the formula for a molecule of sulfur differ.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question : Write the formulas of the following compounds:
(a) lithium carbonate
(b) sodium perchlorate
(c) barium hydroxide
(d) ammonium carbonate
(e) sulfuric acid
(f) calcium acetate
(g) magnesium phosphate
(h) sodium sulfite
Using the periodic table, classify each of the following elements as a metal or a nonmetal, and then further classify each as a main-group (representative) element, transition metal, or inner transition metal:
Write the molecular and empirical formulas of the following compounds:
a)
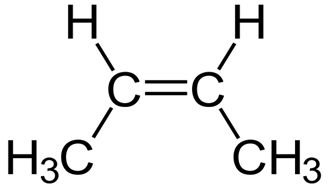
(b)
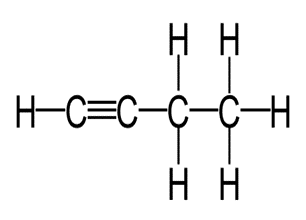
(c)
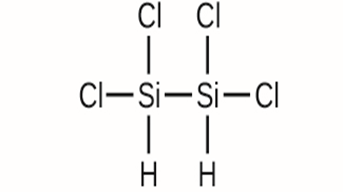
(d)
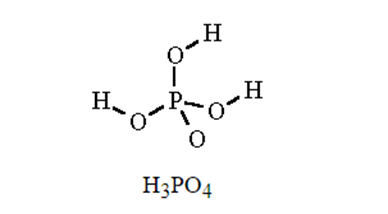
For each of the following compounds, state whether it is ionic or covalent, and if it is ionic, write the symbols for the ions involved:
Each of the following compounds contains a metal that can exhibit more than one ionic charge. Name these compounds:
a. NiCO3
b. MoO3
c.Co(NO3)2
d. V2O5
e. MnO2
f. Fe2O3
What do you think about this solution?
We value your feedback to improve our textbook solutions.