Chapter 2: Q.33E (page 67)
Write the empirical formula for the following compounds:
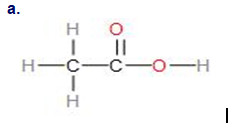
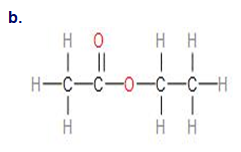
Short Answer
The empirical formula for the given compounds are as follows:
- CH2O
- C2H4O
Learning Materials
Features
Discover
Chapter 2: Q.33E (page 67)
Write the empirical formula for the following compounds:


The empirical formula for the given compounds are as follows:
All the tools & learning materials you need for study success - in one app.
Get started for free
Open theBuild an Atom simulation (http://openstaxcollege.org/l/16PhetAtomBld)and click on the
Atom icon.
a. Pick any one of the first 10 elements that you would like to build and state its symbol.
b. Drag protons, neutrons, and electrons onto the atom template to make an atom of your element.
State the numbers of protons, neutrons, and electrons in your atom, as well as the net charge and mass number.
c. Click on “Net Charge” and “Mass Number,” check your answers to (b), and correct, if needed.
d. Predict whether your atom will be stable or unstable. State your reasoning.
e. Check the “Stable/Unstable” box. Was your answer to (d) correct? If not, first predict what you can do to make a Stable atom of your element, and then do it and see if it works. Explain your reasoning.
The existence of isotopes violates one of the original ideas of Dalton’s atomic theory. Which one?
Each of the following compounds contains a metal that can exhibit more than one ionic charge. Name these compounds:
a. Cr2O3
b. FeCl2
c. CrO3
d. TiCl4
e.CoO
f. MoS2
A sample of magnesium is found to contain 78.70% of 24Mg atoms (mass 23.98 amu), 10.13% of 25Mg atoms (mass 24.99 amu), and 11.17% of 26Mg atoms (mass 25.98 amu). Calculate the average mass of a Mg atom.
Using the periodic table, predict whether the following chlorides are ionic or covalent:\(SiC{l_4},PC{l_3},CaC{l_2},CsCl,CuC{l_2}{\rm{ and }}CrC{l_3}.\)
What do you think about this solution?
We value your feedback to improve our textbook solutions.