Chapter 8: Q43E (page 451)
Compare the atomic and molecular orbital diagrams to identify the member of each of the following pairs that has the highest first ionization energy (the most tightly bound electron) in the gas phase:
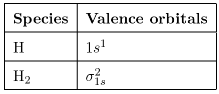
(a) \({\rm{H}}\) and \({{\rm{H}}_{\rm{2}}}\)
(b) \({\rm{N}}\)and \({{\rm{N}}_{\rm{2}}}\)
(c) \({\rm{O}}\)and \({{\rm{O}}_{\rm{2}}}\)
(d) \({\rm{C}}\)and \({{\rm{C}}_{\rm{2}}}\)
(e) \({\rm{B}}\)and \({{\rm{B}}_{\rm{2}}}\)
Short Answer
(a) \({{\rm{H}}_{\rm{2}}}\) has the highest first ionization energy because we need remove an electron from bonding molecular orbital.
(b) \({{\rm{N}}_2}\)has the highest first ionization energy because we need remove an electron from bonding molecular orbital.
(c)O has the highest first ionization energy because we need to remove an electron from antibonding molecular orbital in\({{\rm{O}}_2}\).
(d)\({C_2}\)has the highest first ionization energy because we need to remove an electron from bonding molecular orbital.
(e) \({{\rm{B}}_2}\) has the highest first ionization energy because we need to remove an electron from bonding molecular orbital.