Chapter 8: Q2 CYL (page 423)
What is the hybridization of the selenium atom in SeF4?
Short Answer
The sp3d hybridization of the selenium atom.
Learning Materials
Features
Discover
Chapter 8: Q2 CYL (page 423)
What is the hybridization of the selenium atom in SeF4?
The sp3d hybridization of the selenium atom.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why is the concept of hybridization required in valence bond theory?
For the carbonate ion, \({\rm{C}}{{\rm{O}}_{\rm{3}}}^{{\rm{2 - }}}\), draw all of the resonance structures. Identify which orbitals overlap to create each bond.
If molecular orbitals are created by combining five atomic orbitals from atom A and five atomic orbitals from atom B combine, how many molecular orbitals will result?
For the first ionization energy for an \({{\rm{N}}_2}\) molecule, what molecular orbital is the electron removed from?
For each of the following structures, determine the hybridization requested and whether the electrons will be delocalized:
(a)Hybridization of each carbon

(b)Hybridization of sulphur

(c)
All atoms
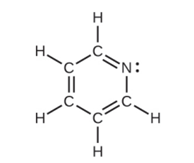
What do you think about this solution?
We value your feedback to improve our textbook solutions.