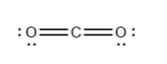Chapter 8: Q29E (page 449)
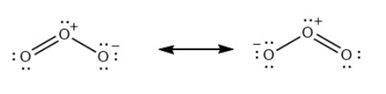
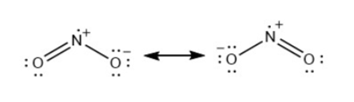
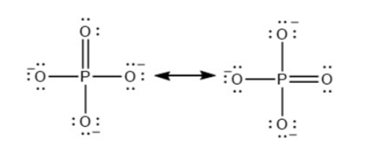
For each of the following molecules, indicate the hybridization requested and whether or not the electrons will be delocalized: (a) ozone (\({{\rm{O}}_{\rm{3}}}\)) central \({\rm{O}}\) hybridization (b) carbon dioxide (\({\rm{C}}{{\rm{O}}_{\rm{2}}}\)) central \({\rm{C}}\) hybridization (c) nitrogen dioxide (\({\rm{N}}{{\rm{O}}_{\rm{2}}}\)) central \({\rm{N}}\) hybridization (d) phosphate ion (\({\rm{P}}{{\rm{O}}_{\rm{4}}}^{{\rm{3 - }}}\)) central \({\rm{P}}\) hybridization.
Short Answer
(a)\({\rm{s}}{{\rm{p}}^{\rm{2}}}\)with electrons delocalized.
(b)\({\rm{sp}}\)with electrons not delocalized.
(c)\({\rm{s}}{{\rm{p}}^{\rm{2}}}\)with electrons delocalized.
(d) \({\rm{s}}{{\rm{p}}^{\rm{3}}}\) with electrons delocalized.