Chapter 15: Q3P (page 412)
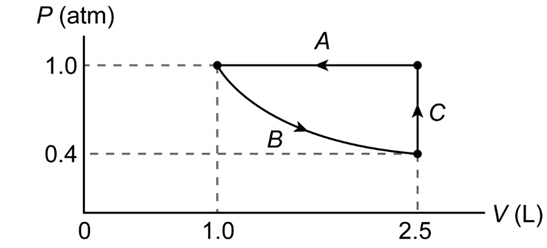
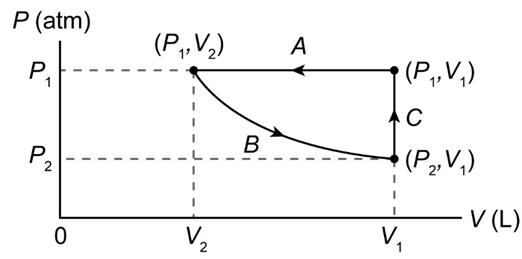
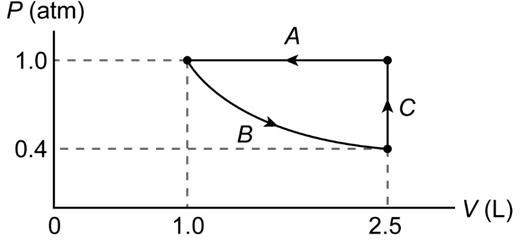
(II) Sketch a PV diagram of the following process: 2.5 L of ideal gas at atmospheric pressure is cooled at constant pressure to a volume of 1.0 L, and then expanded isothermally back to 2.5 L, whereupon the pressure is increased at constant volume until the original pressure is reached.
Short Answer
The PV diagram of the process is shown below.