Chapter 15: Q39P (page 412)
Question: (I) What is the change in entropy of 320 g of steam at 100°C when it is condensed to water at 100°C?
Short Answer
The change in entropy of the steam is \( - 1939\;{\rm{J/K}}\).
Learning Materials
Features
Discover
Chapter 15: Q39P (page 412)
Question: (I) What is the change in entropy of 320 g of steam at 100°C when it is condensed to water at 100°C?
The change in entropy of the steam is \( - 1939\;{\rm{J/K}}\).
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: (II) What volume of water at 0°C can a freezer make into ice cubes in 1.0 h, if the coefficient of performance of the cooling unit is 6.0 and the power input is 1.2 kilowatt?
Question: (II) A 1.0-L volume of air initially at 3.5 atm of (gauge) pressure is allowed to expand isothermally until the pressure is 1.0 atm. It is then compressed at constant pressure to its initial volume, and lastly is brought back to its original pressure by heating at constant volume. Draw the process on a PV diagram, including numbers and labels for the axes.
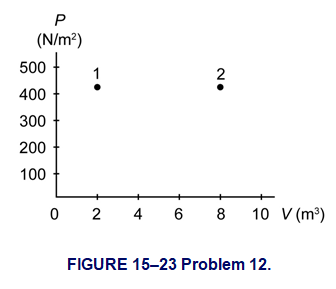
Question: (III) The PV diagram in Fig. 15–23 shows two possible states of a system containing 1.75 moles of a monatomic ideal gas. \(\left( {{P_1} = {P_2} = {\bf{425}}\;{{\bf{N}} \mathord{\left/{\vphantom {{\bf{N}} {{{\bf{m}}^{\bf{2}}}}}} \right.} {{{\bf{m}}^{\bf{2}}}}},\;{V_1} = {\bf{2}}{\bf{.00}}\;{{\bf{m}}^{\bf{3}}},\;{V_2} = {\bf{8}}{\bf{.00}}\;{{\bf{m}}^{\bf{3}}}.} \right)\) (a) Draw the process which depicts an isobaric expansion from state 1 to state 2, and label this process A. (b) Find the work done by the gas and the change in internal energy of the gas in process A. (c) Draw the two-step process which depicts an isothermal expansion from state 1 to the volume \({V_2}\), followed by an isovolumetric increase in temperature to state 2, and label this process B. (d) Find the change in internal energy of the gas for the two-step process B.
A heat engine operates between a high temperature of about 600°C and a low temperature of about 300°C. What is the maximum theoretical efficiency for this engine?
(a) \( = 100\% \). (b) \( \approx 66\% \). (c) \( \approx 50\% \). (d) \( \approx 34\% \).
(e) Cannot be determined from the given information.
Question: Which do you think has the greater entropy, 1 kg of solid iron or 1 kg of liquid iron? Why?
What do you think about this solution?
We value your feedback to improve our textbook solutions.