Chapter 19: Q. 58 (page 545)
of nitrogen gas follow the two processes shown in FIGURE P19.58. How much heat is required for each?
Short Answer
The heat required for isand the heat required forrole="math" localid="1648036368649" is
Learning Materials
Features
Discover
Chapter 19: Q. 58 (page 545)
of nitrogen gas follow the two processes shown in FIGURE P19.58. How much heat is required for each?
The heat required for isand the heat required forrole="math" localid="1648036368649" is
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Is the final temperature higher than, lower than, or equal to the initial temperature?
b. Is any heat energy added to or removed from the system in this process? Explain
The gas cylinder in Figure Q19.10 is well insulated except for the bottom surface, which is in contact with a block of ice.
The piston can slide without friction. The initial gas temperature is
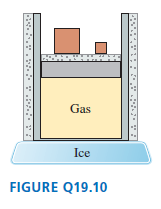
a. During the process that occurs until
the gas reaches a new equilibrium, are
(i) , (ii) W, and (iii) Q greater than, less than, or equal to zero? Explain.
b. Draw a pV diagram showing the process.
A beaker with a metal bottom is filled with of water at . It is brought into good thermal contact with a container holding of a monatomic gas at pressure. Both containers are well insulated from their surroundings.
What is the gas pressure after a long time has elapsed? You can assume that the containers themselves are nearly massless and do not affect the outcome.
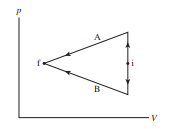
Shows two different processes taking an ideal gas from state i to state f.
Is the work done on the gas in process A greater than, less than, or equal to the work done in process B? Explain.
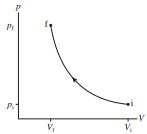
moles of an ideal gas at temperature and volume localid="1648032029846" expand isothermally until the volume has doubled. In terms of and , what are (a) the final temperature, (b) the work done on the gas, and (c) the heat energy transferred to the gas?
What do you think about this solution?
We value your feedback to improve our textbook solutions.