Chapter 39: Q.1. (page 1136)
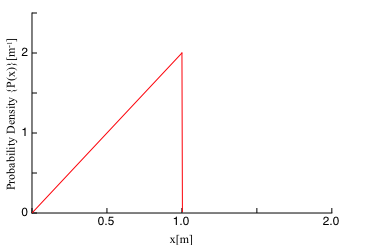
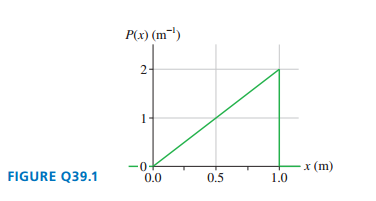
FIGURE Q39.1 shows the probability density for photons to be detected on the-axis.
a. Is a photon more likely to be detected at or at ? Explain.
b. One million photons are detected. What is the expected number of photons in a -wide interval at ?
Short Answer
a. The probability density at wide .
The expected number of detected photon in the interval of at is.